Famoxadone
Synonym(s):3-Anilino-5-methyl-5-(4-phenoxyphenyl)oxazolidine-2,4-dione
- CAS NO.:131807-57-3
- Empirical Formula: C22H18N2O4
- Molecular Weight: 374.39
- MDL number: MFCD03427409
- EINECS: 603-520-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-03 18:03:33
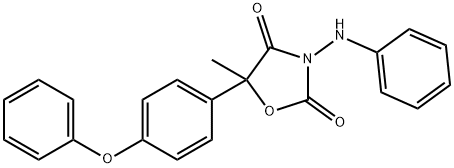
What is Famoxadone?
Description
Famoxadone is an oxazolidinedione fungicide. Although famoxadone is not a strobilurin derivative, it shares the same mechanism of action. Famoxadone provides control of a broad spectrum of fungi but is particularly effective against downy mildew (P. viticola), late and early blights (Phytophthora infestans and Alternaria solani), the Septoria complex, and barley net blotch at rates of 50 to 200 g ai/ha. Target crops include vines, potatoes/tomatoes, cereals, sugar beets, canola, and cucumber. It shows good protectant, translaminar and residual control, with excellent rainfastness and good crop safety. As was seen for trifloxystrobin, it is particularly effective against grape downy mildew when applied in mixture with cymoxanil. The cereal disease spectrum and level of disease control are improved when famoxadone is mixed with triazole fungicides such as flusilazole.
Chemical properties
Brown Solid
The Uses of Famoxadone
Famoxadone is a known fungicide used in the protection of agricultural products against various fungal diseases and other biotic stresses such as pests.
The Uses of Famoxadone
Agricultural fungicide.
The Uses of Famoxadone
Famoxadone provides preventive control of a broad spectrum of fungal pathogens such as Plasmopara viticola (grape downy mildew), Phytophthora infestans (potato/ tomato late blight), Pseudoperonospora cubensis (cucumber downy mildew), Septoria tritici (wheat leaf blotch), S. nodorum (wheat glume blotch) and Alternaria solani (potato/tomato early blight).
Definition
ChEBI: 5-methyl-5-(4-phenoxyphenyl)-3-(phenylamino)-1,3-oxazolidine-2,4-dione is a member of the class of oxazolidinones that is 1,3-oxazolidine-2,4-dione in which the hydrogen attached to the nitrogen is substituted by a phenylamino group and the hydrogens at position 5 are substituted by methyl and 4-phenoxyphenyl groups. It is an aromatic ether, a carbohydrazide and an oxazolidinone.
Agricultural Uses
Fungicide: Tanos 50-DF is recommended for use as a preventative fungicide for control of late blight and early blight on potatoes and field tomatoes.
Trade name
DPX-JE874®; FAMOXATE®; MEDLEY®; TANOS-50DF®
Pharmacology
Famoxadone inhibits activity of ubiquinol : cytochrome c oxidoreductase at the Qo site of complex III, the same target protein as the strobilurins (34,35). However, studies show a difference in the potency of various strobilurins compared with famoxadone in single amino acid mutants of the cytochrome b protein, suggesting that this compound may interact differently with the target protein (38).
Metabolic pathway
Famoxadone undergoes extensive degradation and metabolism in aqueous and soil environments, in wheat plants, rats, goats and hens via common pathways. The primary reactions include the hydrolytic opening of the oxazolidinedione ring, aryl hydroxylation, cleavage of the phenoxyphenyl and the oxazolidinedione-aminophenyl linkages and conjugation. More than 25 degradation products have been identified.
Degradation
Famoxadone is relatively stable to hydrolytic degradation under dark conditions in pH 5 buffer solution (DT50 41 days) (Jernberg et al., 1998a). It was hydrolysed rapidly at pH 7 and pH 9 at 25 °C (DT50 values 2 days and <2 hours, respectively). No sigruficant sunlight-induced degradation was observed at pH 7. Hydrolytic degradation was catalysed in acidic conditions (pH 5) under natural sunlight (DT50 4.6 days in contrast to 41 days at pH 7). Primary hydrolytic degradation reactions include the opening of the oxazolidinedione ring [to yieldα-hydroxy-α-methyl-4-phenoxyphenylacetic acid 2-phenylhydrazide (2) and 1-carboxy-1-(4-phenoxy-phenyl) ethyl-2-phenylhydrazinecarboxylic acid (3)], and the cleavage of the oxazolidine-aminophenyl linkage to yield various products from both the phenoxyphenyl moiety [ 1-(4-phenoxyphenyl)ethanone (4) and α-hydroxy- α-methyl-4-phenoxyphenylacetiac acid (5)] and the aminophenyl moiety [small amounts of phenol (6), catechol (7) and benzene (8) via the proposed 2-phenylhydrazinecarboxylic acid intermediate (9)] (see Scheme 1).
Toxicity evaluation
Famoxadone has an acute oral LD50 > 5,000 mg/kg and an acute percutaneous LD50 > 2,000 mg/kg in rats. It is not a skin or eye irritant, is negative in the Ames test, and is nonteratogenic. It shows very little soil degradation, is nonmobile, and has a good ecotoxicological profile.
Properties of Famoxadone
| Melting point: | 140.3~141.8℃ |
| Boiling point: | 491.3±55.0 °C(Predicted) |
| Density | 1.327±0.06 g/cm3(Predicted) |
| vapor pressure | 6.4 x 10-7 Pa (20 °C) |
| Flash point: | 2 °C |
| storage temp. | 0-6°C |
| solubility | DMSO: 100 mg/mL (267.10 mM) |
| pka | 0.63±0.40(Predicted) |
| Water Solubility | 0.243 mg-1 (pH 5), 0.011 mg l-1 (pH 7)
at 20 °C |
| form | Solid:particulate/powder |
| color | White to yellow |
| EPA Substance Registry System | Famoxadone (131807-57-3) |
Safety information for Famoxadone
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H225:Flammable liquids H302:Acute toxicity,oral H312:Acute toxicity,dermal H319:Serious eye damage/eye irritation H332:Acute toxicity,inhalation |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Famoxadone
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran


You may like
-
 Famoxadone 98% CAS 131807-57-3View Details
Famoxadone 98% CAS 131807-57-3View Details
131807-57-3 -
 Famoxadone solution CAS 131807-57-3View Details
Famoxadone solution CAS 131807-57-3View Details
131807-57-3 -
 Famoxadone CAS 131807-57-3View Details
Famoxadone CAS 131807-57-3View Details
131807-57-3 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 -
 4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
5305-40-8
