Ethynodiol diacetate
Synonym(s):(3β, 17α)-19-Norpregn-4-en-20-yne-3,17-diol diacetate;3β,17β-Diacetoxy-17α-ethynyl-4-estrene;Etynodiol acetate
- CAS NO.:297-76-7
- Empirical Formula: C24H32O4
- Molecular Weight: 384.51
- MDL number: MFCD00083326
- EINECS: 206-044-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-11-03 16:33:47
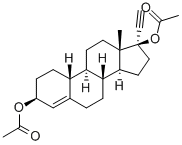
What is Ethynodiol diacetate?
Description
Ethynodiol diacetate is a synthetic progestogen. It prevents the development of osteoporosis in ovariectomized rats when administered at a dose of 9 μg/animal per day. Ethynodiol diacetate inhibits copulatory ovulation in rabbits. Formulations containing ethynodiol diacetate have been used as oral contraceptives.
Originator
Lutometrodiol,Searle,France,1965
The Uses of Ethynodiol diacetate
Ethynodiol Diacetate is used as a hormonal contraceptive, birth control.
The Uses of Ethynodiol diacetate
anticonvulsant
Indications
For the prevention of pregnancy in women who elect to use this product as a method of contraception.
Background
A synthetic progestational hormone used alone or in combination with estrogens as an oral contraceptive. Although etynodiol or ethynodiol are sometimes used as a synonym for ethynodiol diacetate, what is usually being referred to is actually ethynodiol diacetate and not ethynodiol (which is a separate drug that has never been marketed, see Etynodiol).
Definition
ChEBI: Ethynodiol diacetate is a steroid ester and a terminal acetylenic compound. It has a role as an estrogen receptor modulator, a contraceptive drug and a synthetic oral contraceptive. It is functionally related to an ethynodiol.
Manufacturing Process
A mixture of 30 parts of 17α-ethynyl-19-norandrost-4-ene-3β,17β-diol, 360 parts of dry pyridine, and 111 parts of acetic anhydride, under nitrogen, is stirred and heated at the reflux temperature for about 5 hours. This reaction mixture is cooled, then poured into approximately 3,500 parts of cold water and the resulting aqueous mixture is stirred at room temperature for about 0.5 hour. The precipitate which forms is collected by filtration, then is washed on the filter with water and dried in air. This solid material is extracted into ether, and the ether solution is washed successively with 10% aqueous
Therapeutic Function
Progestin; Oral contraceptive ingredient
General Description
Ethynodiol diacetate, 19-norpregn-4-en-20-yne-3β,17α-diol diacetate, is a prodrug ofnorethindrone. A combination of hydrolysis of both estersand oxidation of the C3 alcohol to the ketone is necessary toprovide the fully active progestin.
Pharmacokinetics
Ethynodiol Diacetate is used as a female contraceptive. Ethynodiol Diacetate is a progestin or a synthetic form of the naturally occurring female sex hormone, progesterone. In a woman's normal menstrual cycle, an egg matures and is released from the ovaries (ovulation). The ovary then produces progesterone, preventing the release of further eggs and priming the lining of the womb for a possible pregnancy. If pregnancy occurs, progesterone levels in the body remain high, maintaining the womb lining. If pregnancy does not occur, progesterone levels in the body fall, resulting in a menstrual period. Ethynodiol Diacetate tricks the body processes into thinking that ovulation has already occurred, by maintaining high levels of the synthetic progesterone. This prevents the release of eggs from the ovaries.
Safety Profile
Suspected carcinogen. Human reproductive effects by ingestion: menstrual cycle changes. Experimental reproductive effects. Mutation data reported. A steroid. When heated to decomposition it emits acrid smoke and irritating fumes.
Metabolism
Not Available
Properties of Ethynodiol diacetate
| Melting point: | ~126-127° |
| Boiling point: | 431.17°C (rough estimate) |
| alpha | D -72.5° (chloroform) |
| Density | 1.0474 (rough estimate) |
| refractive index | 1.4860 (estimate) |
| storage temp. | 2-8°C |
| solubility | Chloroform (Slightly), DMSO (Slightly) |
| form | Solid |
| color | White |
| Water Solubility | 1.4mg/L(25 ºC) |
| CAS DataBase Reference | 297-76-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Ethynodiol diacetate(297-76-7) |
| EPA Substance Registry System | Ethynodiol diacetate (297-76-7) |
Safety information for Ethynodiol diacetate
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H351:Carcinogenicity |
| Precautionary Statement Codes |
P201:Obtain special instructions before use. P280:Wear protective gloves/protective clothing/eye protection/face protection. P308+P313:IF exposed or concerned: Get medical advice/attention. |
Computed Descriptors for Ethynodiol diacetate
Abamectin manufacturer
Ralington Pharma
New Products
Tubulysin H Tubulysin F Tubulysin I 2,2-diethoxyethanethioamide 4,4-DIFLUOROCYCLOHEXANAMINE 3-FLUOROPYRROLIDINE HYDROCHLORIDE 1-Boc-5-broMo-1H-pyrazolo[3,4-b]pyridine 4,4-DIFLUOROCYCLOHEXYLAMINE HYDROCHLORIDE 4-CYANO-TETRAHYDROPYRAN-4-CARBOXYLIC ACID 3,6-Dihydro-2H-pyran-4-carbonitrile (S)-4-(azidomethyl) -3-(3-fluoro-4- thiomorpholino phenyl) oxazolidin-2-one (+/-)-6-Acetylamino-2- amino-4, 5, 6, 7-tetrahydro Benzo thiazole (R)-1-Aminoindan Hydrochloride 2,3-Dichloro-6- nitrobenzylamine Hydrochloride 5-(piperazin-1-yl) benzofuran-2- carboxamide 2-(2-Formylphenoxy) acetic acid 7-Methoxyquinoline-4-carboxylic acid 7-Methoxyquinoline-4-carbonitrile 1H-Imidazole-4-carbonitrile (R)-methyl 2-isocyano-3-(tritylthio)propanoate (R)-methyl 2-formamido-3-(tritylthio)propanoate Montelukast 1,2-Diol / Montelukast 1,2-Diol (Mixture of Diastereomers) BECLOMETASONE DIPROPIONATE EP IMPURITY N Montelukast (3S)-Hydroxy BenzoateRelated products of tetrahydrofuran

You may like
-
 297-76-7 Ethynodiol diacetate 98%View Details
297-76-7 Ethynodiol diacetate 98%View Details
297-76-7 -
 Ethynodiol Diacetate CAS 297-76-7View Details
Ethynodiol Diacetate CAS 297-76-7View Details
297-76-7 -
 Ethynodiol diacetate CAS 297-76-7View Details
Ethynodiol diacetate CAS 297-76-7View Details
297-76-7 -
 Ethynodiol diacetate 98.00% CAS 297-76-7View Details
Ethynodiol diacetate 98.00% CAS 297-76-7View Details
297-76-7 -
 Ethynodiol diacetate >98% (HPLC) CAS 297-76-7View Details
Ethynodiol diacetate >98% (HPLC) CAS 297-76-7View Details
297-76-7 -
 3-(4-Chloro-butyryl) -1H-indole-5- carbonitrileView Details
3-(4-Chloro-butyryl) -1H-indole-5- carbonitrileView Details
276863-95-7 -
 Ethyl 5-Aminobenzofuran-2- CarboxylateView Details
Ethyl 5-Aminobenzofuran-2- CarboxylateView Details
174775-48-5 -
 2,3-dichloro-6- nitrobenzonitrileView Details
2,3-dichloro-6- nitrobenzonitrileView Details
2112-23-2