Dimethylamine
Synonym(s):DMA
- CAS NO.:124-40-3
- Empirical Formula: C2H7N
- Molecular Weight: 45.08
- MDL number: MFCD00008288
- EINECS: 204-697-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-09-25 17:15:13
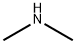
What is Dimethylamine?
Description
Dimethylamine is a colourless flammable gas at room temperature. It has a pungent, fishy, or ammonia-like odour at room temperature and is shipped and marketed in compressed liquid form. It is very soluble in water and soluble in alcohol and ether. It is incompatible with oxidising materials, acrylaldehyde, fluorine, maleic anhydride, chlorine, or mercury. Dimethylamine is a precursor to several industrially important compounds. For instance, it used in the manufacture of several products, for example, for the vulcanisation process of rubber, as detergent soaps, in leather tanning, in the manufacture of pharmaceuticals, and also for cellulose acetate rayon treatment.
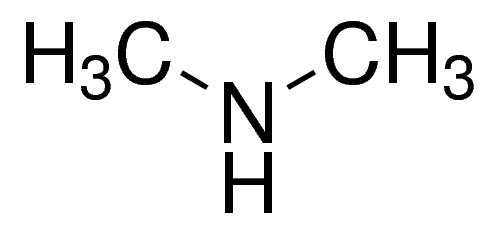
dimethylamine structure
Chemical properties
Dimethylamine reacts readily with acids to produce salts due to the presence of the unshared electron pair on the nitrogen atom. Similarly, dimethylamine reacts with acid anhydrides, halides, and esters, with CO2 or CS2, or with isocyanic or isothiocyanic acid derivatives. It can also react with nitrite, especially under acidic conditions, and possibly nitrogen oxides (Iqbel 1986) to form N-nitrosodimethylamine, a potent carcinogen in various animal species and a suspect human carcinogen (ATSDR 1989; Scanlan 1983; Zeisel et al 1988). N-Nitrosodimethylamine also can be formed upon storage of aqueous dimethylamine solutions or formulations of the dimethylamine salts of the herbicides 2,4D and MCPA (Wigfield and McLenaghan 1987a,b). Dimethylamine also can be nitrosated photochemically in aqueous solutions containing nitrite with the reaction occurring most readily at alkaline pH (Ohta et al 1982).
Physical properties
Clear, colorless liquid or gas with a strong, ammonia-like odor. Odor threshold concentrations of 33 ppbv and 47 ppbv were experimentally determined by (Leonardos et al., 1969) and Nagata and Takeuchi (1990), respectively.
The Uses of Dimethylamine
Dimethylamine is a used as a precursor to several industrially significant compounds. It reacts with carbon disulfide to give dimethyl dithiocarbamate, a precursor to a family of chemicals widely used in the vulcanization of rubber. It is raw material for the production of many agrochemicals and pharmaceuticals, such as dimefox and diphenhydramine, respectively.
The Uses of Dimethylamine
Dimethylamine is used in the manufactureof N-methylformamide, N-methylacetamide,and detergent soaps; in tanning; and as anaccelerator in vulcanizing rubber. It is commercially sold as a compressed liquid intubes or as a 33% aqueous solution..
The Uses of Dimethylamine
Manufacture of pharmaceuticals; stabilizer in gasoline; in production of insecticides and fungicides; in manufacture of soaps and surfactants
Definition
ChEBI: A secondary aliphatic amine where both N-substituents are methyl.
Production Methods
Methods used commercially for the large-scale production of dimethylamine are
generally those used for methylamine synthesis (HSDB 1989). The most widely
used process involves heating ammonium chloride and methyl alcohol to about
300°C in the presence of a dehydrating catalyst such as zinc chloride. Dimethylamine
has also been prepared from methanol and ammonia or by the catalytic
hydrogenation of nitrosodimethylamine (Schweizer et al 1978). It is usually
marketed in compressed liquid (anhydrous) form or as a 25-60% aqueous solution.
Dimethylamine is also naturally present in biological systems, probably being
formed as a breakdown product from trimethylamine N-oxide (Timofievskaja
1984). Thus it is present in gastric juice of humans, rats, dogs and ferrets at
concentrations of 12.6 ± 14 nmol/ml (Zeisel et al 1988); it is a constituent of most
foods, especially seafood including squid and octopus, frequently eaten in traditional
Chinese and Japanese diets, where it reaches concentrations of 946-2043
p.p.m. (Lin et al 1983,1984). Food processing and cooking markedly increases the
dimethylamine contents of foods by increasing the breakdown of constituents such
as trimethylamine N-oxide and sarcosine (Lin et al 1983, 1984; Lin and Hurng
1985). Dimethylamine occurs in the air of iron foundries where the amine was
used in the casting process (Hansen et al 1985) and also is released from plastic
material used in construction (Kiselev et al 1983).
Nitrosation of dimethylamine occurs forming the carcinogenic N-nitrosodimethylamine
upon storage of anhydrous and aqueous solutions of dimethylamine
or formulations of the dimethylamine salts of the herbicides 2,4-dichlorophenoxyacetic
acid (2,4D), 4-chloro-2-methylphenoxyacetic acid (MCPA) and 3,6-dichloro-
2-methoxybenzoic acid (dicamba) (Wigfield and McLenaghan 1987a,b).
The volatile N-nitrosodimethylamine is also formed in foods by reaction of
dimethylamine with sodium nitrite added as a preservative or by reaction with
atmospheric nitrogen oxides during food processing (ATSDR 1989; Gross and
Newberne 1977; Scanlan 1983). Concentrations of the nitrosoamine in cheese,
apple cider, milk, cereals, vegetables, seafood, cured meats, etc. range between
0.05 and 130 p.p.b. (ATSDR 1989).
Air & Water Reactions
Highly flammable. Water soluble.
Reactivity Profile
DIMETHYLAMINE is a base, neutralizing acids in exothermic reactions, and a reducing agent. Dimethylamine is temperature sensitive. Reacts vigorously with mercury and chlorine . Reacts violently with strong oxidizing agents and attacks copper and copper compounds [Handling Chemicals Safely, 1980 p. 123]. Reacts with hypochlorites to give N-chloroamines, some of which are explosives when isolated [Bretherick, 1979 p. 108].
Hazard
Dimethylamine is an irritant, with a TLV of 10 ppm in air. The four-digit UN identification number is 1032. The NFPA 704 designation is health 3, flammability 4, and reactivity 0. The primary uses are in electroplating and as gasoline stabilizers, pharmaceuticals, missile fuels, pesticides, and rocket propellants.
Health Hazard
Dimethylamine is a strong irritant to the eyes,skin, and mucous membranes. Spill of liquidinto the eyes can cause corneal damage andloss of vision. Skin contact with the liquidcan produce necrosis. At sublethal concentra tions, inhalation of dimethylamine producedrespiratory distress, bronchitis, pneumonitis,and pulmonary edema in test animals. Theacute oral toxicity was moderate, greater thanfor monomethylamine.
LC50 value, inhalation (rats): 4540 ppm/6 hLD50 value, oral (mice): 316 mg/kg
Buckley and coworkers (1985) have investigated the inhalation toxicity of dimethylamine in F-344 rats and B6C3F1 mice.Animals exposed to 175 ppm for 6 h/day,5 days/week for 12 months showed significant lesions in the nasal passages. Rats developed more extensive olfactory lesions thandid mice. The study indicated that olfactory sensory cells were highly sensitive todimethylamine. Even at a concentration of10 ppm, the current threshold limit value,the rodents developed minor lesions fromexposure.
Fire Hazard
FLAMMABLE. Flashback along vapor trail may occur. May explode if ignited in an enclosed area. Vapors are eye, skin and respiratory irritants.
Flammability and Explosibility
Extremely flammable liquified gas
Industrial uses
Dimethylamine is used as an accelerator in vulcanizing rubber, as an antiknock agent for fuels, in photography, as a plasticizer, ion exchange agent, as an acid gas absorbent, a flotation agent, a dehairing agent in the tanning of leather and in electroplating (HSDB 1989; Sax and Lewis 1987; Windholz et al 1983). Dimethylamine also serves as the base for a large number of commercial products including detergent soaps, dyes, pharmaceuticals, textile chemicals, surfactants and in the manufacture of unsymmetrical dimethylhydrazine (used in missile fuels), the solvent dimethylacetanilide and in the synthesis of dimethylformamide, one of the most commonly used organic solvents. Usage of dimethylamine in 1972 was estimated at 50% for production of dimethylformamide and dimethylacetamide (used as spinning solvents for acrylic fibers), 15% as an intermediate in the preparation of the surfactant laurel dimethylamine oxide, 15% as an intermediate for rubber chemicals (including thorium accelerators), and 20% for other applications including the production of unsymmetrical dimethylhydrazine in rocket fuels and the dimethylamine salt of 2,4-dichlorophenoxyacetic acid (HSDB 1989). U.S. production and sales of dimethylamine in 1985 was 65.9 million pounds.
Safety Profile
Poison by ingestion. Moderately toxic by inhalation and intravenous routes. Mutation data reported. An eye irritant. Corrosive to the eyes, skin, and mucous membranes. A flammable gas. When heated to decomposition it emits toxic fumes of Nx,. Incompatible with acrylddehyde, fluorine, and maleic anhydride
Potential Exposure
Mutagen.Primary Irritant. This material is used in leather tanning; asan accelerator in rubber vulcanization; in the manufactureof detergents; in drug synthesis and pesticide manufacture.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediately with soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.Medical observation is recommended for 24-48 h afterbreathing overexposure, as pulmonary edema may bedelayed. As first aid for pulmonary edema, a doctor orauthorized paramedic may consider administering a corticosteroid spray.If frostbite has occurred, seek medical attention immediately; do NOT rub the affected areas or flush them withwater. In order to prevent further tissue damage, do NOTattempt to remove frozen clothing from frostbitten areas. Iffrostbite has NOT occurred, immediately and thoroughlywash contaminated skin with soap and water.
Carcinogenicity
In a 2 year inhalation study in male F344 rats exposed to
175 ppm, no evidence of carcinogenicity was observed, and
in addition, despite severe tissue destruction in the anterior
nose following a single 6 h exposure, the nasal lesions
exhibited very little evidence of progression, even at
2 years of exposure. The authors concluded that this
indicated possible regional susceptibility to DMA toxicity or
a degree of adaptation by the rat to continued DMA exposure.
A detailed evaluation of mucociliary apparatus function
and response to alterations of nasal structure was presented
by the authors.
Source
Dimethylamine naturally occurs in soybean seeds (8 ppm), cauliflower (14 ppm), kale leaves (5.5 ppm), barleygrass seeds (1.6 ppm), tobacco leaves, hawthorne leaves, hops flower (1.4 ppm), cabbage leaves (2–2.8 ppm), corn (1–3.5 ppm), celery (5.1 ppm), grapes, grape wine, and grape juice (Duke, 1992).
Environmental Fate
Photolytic. Dimethylnitramine, nitrous acid, formaldehyde, N,N-dimethylformamide and carbon
monoxide were reported as photooxidation products of dimethylamine with NOx. An additional
compound was tentatively identified as tetramethylhydrazine (Tuazon et al., 1978). In the
atmosphere, dimethylamine reacts with OH radicals forming formaldehyde and/or amides
(Atkinson et al., 1978). The rate constant for the reaction of dimethylamine and ozone in the
atmosphere is 2.61 x 10-18 cm3/molecule?sec at 296 K (Atkinson and Carter, 1984).
Soil. After 2 d, degradation yields in an Arkport fine sandy loam (Varna, NY) and sandy soil
(Lake George, NY) amended with sewage and nitrite-N were 50 and 20%, respectively. NNitrosodimethylamine
was identified as the major metabolite (Greene et al., 1981). Mills and
Alexander (1976) reported that N-nitrosodimethylamine also formed in soil, municipal sewage,
and lake water supplemented with dimethylamine (ppm) and nitrite-N (100 ppm). They found that
nitrosation occurred under nonenzymatic conditions at neutral pHs.
Photolytic. Low et al. (1991) reported that the photooxidation of aqueous secondary amine
solutions by UV light in the presence of titanium dioxide resulted in the formation of ammonium
and nitrate ions.
Chemical/Physical. In an aqueous solution, chloramine reacted with dimethylamine forming
N-chlorodimethylamine (Isaac and Morris, 1983).
Reacts with mineral acids forming water soluble ammonium salts and ethanol (Morrison and
Boyd, 1971).
Metabolism
Dimethylamine is normally present in the stomach and urine of animals and
humans. The secondary amine is formed from trimethylamine (a breakdown
product of dietary choline) via trimethylamine N-oxide (Zeisel et al 1985) and
probably also from dietary lecithin and creatine (Lewis et al 1985). Enzymes
within gut bacteria catalyze these conversions. The resulting dimethylamine is
readily absorbed primarily from the small intestine, and to a much lesser extent,
the stomach, and excreted in the urine (Ishiwata et al 1984; Zeisel et al 1983).
Humans consuming a diet high in fish show at least a 4-fold increase in urinary
dimethylamine excretion (Zeisel and Dacosta 1986).
Although dimethylamine may arise primarily from trimethylamine in a process
catalyzed by bacteria, when rats were fed a commercial diet containing 23.6 p.p.m.
dimethylamine, nearly 50% of the amine was recovered in the stomach with
progressively declining amounts found towards lower regions of the gastrointestinal
tract (Ishiwata et al 1984). Using ligated sections, the t1/2 of dimethylamine
was found to be 198 min in the stomach with the intestines and caecum varying
from 8.3-31.5 min. The results indicated that dimethylamine is rapidly absorbed
from the intestine and into the blood from where it disappears quickly, to be
excreted predominately in the urine with a small amount excreted into the bile.
In rats fed a choline deficient diet, or rats devoid of gut bacteria, dimethylamine
was still excreted in the urine (Zeisel et al 1985). This suggests that mammalian
cells may possess other, as yet undefined, endogenous pathways for forming
dimethylamine. The absorption, distribution and secretion of dimethylamine in the
digestive tract and its biliary and urinary excretion was studied in male Wistar rats
(Ishiwata et al 1984). Animals were fed diets containing 1 or 23.6 p.p.m.
dimethylamine for one wk and then killed. Single i.v. doses also were administered
to control and bile-duct cannulated rats and the urine collected over a 24 h
period. The authors found high dimethylamine concentrations in the upper part of
the gastrointestinal tract and a low concentration in the lower intestine. The
half-life for injected dimethylamine was 12.5 min and excretion was primarily via
the bile.
The disposition and pharmacokinetics of [14C]-dimethylamine were also studied
in male Fischer 344 rats following 6 h inhalation of 10 or 175 p.p.m. of the labeled
amine (McNulty and Heck 1983). At 72 h after exposure, the disposition at both
doses was similar with greater than 90% of the radioactivity appearing in the urine
and feces, 7-8% in various tissues and 1.5% exhaled as 14CO2. Over 98% of the
urinary radioactivity was the parent [14C]-dimethylamine. However, some formation
of small quantities of dimethylamine oxidative metabolites was seen.
Much of the concern over the presence of dimethylamine in humans stems from
its ability to serve as a precursor for the formation of the putative carcinogen,
N-nitrosodimethylamine. Accordingly, several studies have been conducted to
assess the potential for exogenously administered dimethylamine to form this
nitroso compound. When dimethylamine was given intravenously to dogs and
ferrets, the amine was rapidly transported from the blood into the gastric fluid,
where N-nitrosodimethylamine formation can occur (Zeisel et al 1986). Nnitrosodimethylamine
was formed in vitro when sodium nitrite was added to dog
(Lintas et al 1982) or human gastric fluid (Zeisel et al 1988). The resulting
N-nitrosamine then is rapidly absorbed from the stomach. When conventional and
germfree male Wistar rats were treated with dimethylamine and sodium nitrite,
severe liver necrosis was observed at 48 h only in the germfree animals (Sumi and
Miyakawa 1983). This may indicate, at least in this species, that metabolism of
dimethylamine by intestinal microflora may minimize nitrosamine formation.
7V-nitrosodimethylamine requires metabolic activation to form the reactive alkylating
species responsible for the carcinogenic and mutagenic activity of the
nitrosamine (ATSDR 1989).
Storage
Dimethylamine should be stored in a cool, dry, well-ventilated area in tightly sealed containers that are labeled in accordance with OSHA’s Hazard Communication Standard [29 CFR 1910.1200]. Containers of dimethylamine should be protected from physical damage and ignition sources, and should be stored separately from oxidizing materials, acrylaldehyde, fl uorine, maleic anhydride, chlorine, and mercury. Outside or detached storage is preferred. If stored inside, a standard flammable liquids cabinet or room should be used. Ground and bond metal containers and equipment when transferring liquids. Empty containers of dimethylamine should be handled appropriately.
Shipping
This compound requires a shipping label of“POISON GAS, FLAMMABLE GAS.” It falls in HazardClass 2.1.
Purification Methods
Dry dimethylamine by passage through a KOH-filled tower, or by standing with sodium pellets at 0o during 18hours. [Beilstein 4 IV 128.]
Incompatibilities
Dimethylamine is a medium strong base.Reacts violently with strong oxidizers; with mercury causing fire and explosion hazard. Incompatible with acids,organic anhydrides, isocyanates, vinyl acetate, acrylates,substituted allyls, alkylene oxides, epichlorohydrin, ketones,aldehydes, alcohols, glycols, phenols, cresols, caprolactumsolution. Attacks aluminum, copper, lead, tin, zinc andalloys, some plastics, rubbers, and coatings.
Precautions
During handling of dimethylamine, workers should use proper fume hoods, personal protective clothing and equipment, avoid skin contact, and use gloves, sleeves, and encapsulating suits. Dimethylamine is extremely flammable and may be ignited by heat, sparks, or open flames. Liquid dimethylamine will attack some forms of plastic, rubber, and coatings and is flammable. The vapors of dimethylamine are an explosion and poison hazard. Containers of dimethylamine may explode in the heat of a fi re and require proper disposal. Workers should use dimethylamine with adequate ventilation and containers must be kept properly closed.
Properties of Dimethylamine
| Melting point: | −93 °C(lit.) |
| Boiling point: | 7 °C(lit.) |
| Density | 0.89 g/mL at 25 °C |
| vapor density | 1.55 (vs air) |
| vapor pressure | 16.97 psi ( 55 °C) |
| refractive index | n |
| Flash point: | 60 °F |
| storage temp. | Flammables area |
| solubility | very soluble in water (163 g/100 g water at 40°C); soluble in ethanol,
ethyl ether, and many organic solvents |
| form | Solution |
| pka | 10.68(at 25℃) |
| color | Clear slightly yellow |
| Odor | Ammonical odour |
| explosive limit | 14.4% |
| Odor Threshold | 0.033ppm |
| Water Solubility | Miscible with water and most organic solvents. |
| Sensitive | Hygroscopic |
| Merck | 14,3228 |
| BRN | 605257 |
| Henry's Law Constant | 1.75(x 10-5 atm?m3/mol) at 25 °C (Christie and Crisp, 1967) |
| Dielectric constant | 6.3(0℃) |
| Exposure limits | TLV-TWA 10 ppm (~18 mg/m3) (ACGIH,
MSHA, and OSHA); IDLH 2000 ppm
(NIOSH). |
| Stability: | Stable. Generally used as a solution in water at concentrations up to around 40%. Extremely flammable in the pure form. Incompatible with strong oxidizing agents. |
| CAS DataBase Reference | 124-40-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Methanamine, N-methyl-(124-40-3) |
| EPA Substance Registry System | Dimethylamine (124-40-3) |
Safety information for Dimethylamine
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H225:Flammable liquids H314:Skin corrosion/irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation H412:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P233:Keep container tightly closed. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Dimethylamine
Dimethylamine manufacturer
JSK Chemicals
Sainor Laboratories Pvt Ltd Unit III
Belami Fine Chemicals Private Limited
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
You may like
-
 Di Methyl Amine 99%View Details
Di Methyl Amine 99%View Details -
 Dimethylamine 98%View Details
Dimethylamine 98%View Details -
 Dimethylamine CAS 124-40-3View Details
Dimethylamine CAS 124-40-3View Details
124-40-3 -
 Dimethylamine CAS 124-40-3View Details
Dimethylamine CAS 124-40-3View Details
124-40-3 -
 Dimethylamine CASView Details
Dimethylamine CASView Details -
 Dimethylamine CASView Details
Dimethylamine CASView Details -
 Dimethyl Amine (Dimethylamine) - CAS: 124-40-3, Grade Standard: Chemical GradeView Details
Dimethyl Amine (Dimethylamine) - CAS: 124-40-3, Grade Standard: Chemical GradeView Details
124-40-3 -
 Industrial Grade Dimethylamine 40% (DMA 40%), 99%, LiquidView Details
Industrial Grade Dimethylamine 40% (DMA 40%), 99%, LiquidView Details
124-40-3
