Diamylamine
- CAS NO.:2050-92-2
- Empirical Formula: C10H23N
- Molecular Weight: 157.3
- MDL number: MFCD00009499
- EINECS: 218-108-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-09-25 17:15:13
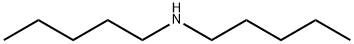
What is Diamylamine?
Chemical properties
colourless liquid
Chemical properties
Diamylamine is a relatively strong base and forms salts with acids. Its vapors can form explosive mixtures with air.
The Uses of Diamylamine
Di-n-amylamine is manufactured from amyl chloride and ammonia. It is used in organic syntheses and as a solvent, rubber accelerator, flotation reagent, and corrosion inhibitor.
The Uses of Diamylamine
Dipentylamine was used in the synthesis of new melt-spinnable polymeric precursor to boron nitride ceramic fibers. It was used to compose background electrolyte for the separation of linear alkylbenzene sulfonates by nonaqueous capillary electrophoresis. It was employed as organic additive in the synthesis of pure AlP04-H2 (aluminophosphate material).
Production Methods
Diamylamine is manufactured by the same processes as n-amylamine by reaction of amyl chloride with ammonia and then separated from the amylenes and amyl alcohol by steam distillation (Hawley 1977). It also can be synthesized by amination of alkyl halides at high temperature and pressure (Schweizer et al 1978). The commercial product may be a mixture of amyl isomers (HSDB 1989).
General Description
A clear colorless liquid with an ammonia-like odor. Very slightly soluble in water. Density 6.40 lb / gal (less than water) Vapors heavier than air. Flash point 152°F. Difficult to ignite. Moderately toxic. Contact with liquid may cause a chemical burn. Vapors may irritate respiratory tract. Used in the manufacture of rubber, resins, and dyes.
Air & Water Reactions
Flammable. Sensitive to air and heat. Slightly soluble in water.
Reactivity Profile
Diamylamine neutralizes acids to form salts plus water. May be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated in combination with strong reducing agents, such as hydrides.
Health Hazard
TOXIC; may be fatal if inhaled, ingested or absorbed through skin. Inhalation or contact with some of these materials will irritate or burn skin and eyes. Fire will produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution.
Health Hazard
Diamylamine is a strong eye, skin, and respiratory irritant owing to its basicity (HSDB 1989). Vapor exposure results in irritation of the nose and throat with distressed breathing and coughing. Prolonged exposure may lead to pulmonary edema. Direct skin contact can cause secondary burns.
Fire Hazard
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion and poison hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
Industrial uses
Diamylamine is less widely used than n-amylamine with only 20 tons being manufactured in the U.S. in 1976. Its most widespread use is as a corrosion inhibitor and rubber accelerator (Hawley 1977). It is also useful as a solvent for oils, resins, and some cellulose esters. Introduction of the amyl group imparts oil solubility to otherwise oil-insoluble substances. Diamylamine also is used in flotation reagents, dyestuffs and as a cockroach repellent (HSDB 1989).
Safety Profile
Poison by inhalation, ingestion, and skin contact. A severe skin irritant. See also AMINES. Flammable liquid when exposed to heat or flame; can react with oxidizing materials. To fight fire, use alcohol foam, foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of NOx.
Metabolism
In contrast to n-amylamine, little information is available on diamylamine metabolism,
particularly with respect to its suitability as a substrate for the amine
oxidases. Generally, the rate of oxidation of secondary amines by monoamine
oxidase is slower than that of primary amines (Beard and Noe 1981). In agreement,
Yamada et al (1965) demonstrated that crystalline amine oxidase prepared
from Aspergillus niger oxidized diamylamine very slowly with respect to n-amylamine.
As with other secondary aliphatic amines, the propensity of diamylamine to
form nitrosamines is of interest. It has been shown that treatment of diamylamine
with nitrous acid in dilute aqueous solution gave optimum nitrosamine formation
between pH 1 and 3, corresponding to stomach conditions (Sander et al 1968).
When rats were fed a diet supplemented with sodium nitrite and secondary amines
of low basicity, synthesis of nitrosamines in the stomach was observed. Malignant
tumors arising through formation of nitrosamines in the stomach was demonstrated
only when nitrite was present in the stomach concomitantly with secondary
amines which readily formed carcinogenic nitrosamines.
Properties of Diamylamine
| Melting point: | -44°C |
| Boiling point: | 202-203 °C(lit.) |
| Density | 0.767 g/mL at 25 °C |
| vapor density | 5.42 (vs air) |
| vapor pressure | 0.3 mm Hg ( 20 °C) |
| refractive index | n |
| Flash point: | 157 °F |
| form | clear liquid |
| pka | 11.25±0.19(Predicted) |
| color | Colorless to Light yellow to Light orange |
| Specific Gravity | 0.775 (25/4℃) |
| Water Solubility | Slightly soluble (0.1-1 g/100 mL) |
| BRN | 906746 |
| Dielectric constant | 2.5 |
| Stability: | Stable, but air-sensitive. Flammable. Incompatible with oxidizing agents. |
| CAS DataBase Reference | 2050-92-2(CAS DataBase Reference) |
| NIST Chemistry Reference | 1-Pentanamine, N-pentyl-(2050-92-2) |
| EPA Substance Registry System | Diamylamine (2050-92-2) |
Safety information for Diamylamine
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Corrosion Corrosives GHS05  Skull and Crossbones Acute Toxicity GHS06  Environment GHS09 |
| GHS Hazard Statements |
H226:Flammable liquids H314:Skin corrosion/irritation H400:Hazardous to the aquatic environment, acute hazard |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P233:Keep container tightly closed. P240:Ground/bond container and receiving equipment. P260:Do not breathe dust/fume/gas/mist/vapours/spray. P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P310:Immediately call a POISON CENTER or doctor/physician. P301+P310:IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P370+P378:In case of fire: Use … for extinction. P405:Store locked up. P403+P235:Store in a well-ventilated place. Keep cool. P501:Dispose of contents/container to..… |
Computed Descriptors for Diamylamine
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran

You may like
-
 Diamylamine (mixture) CAS 2050-92-2View Details
Diamylamine (mixture) CAS 2050-92-2View Details
2050-92-2 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 614-19-7 98%View Details
614-19-7 98%View Details
614-19-7 -
 3112-85-4 Methyl phenyl sulfone 98%View Details
3112-85-4 Methyl phenyl sulfone 98%View Details
3112-85-4 -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 -
 4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
5305-40-8
