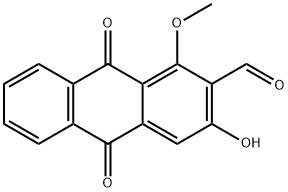
DAMNACANTHAL
Synonym(s):3-Hydroxy-1-methoxyanthraquinone-2-aldehyde;Damnacanthal - CAS 477-84-9 - Calbiochem
- CAS NO.:477-84-9
- Empirical Formula: C16H10O5
- Molecular Weight: 282.25
- MDL number: MFCD00674209
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-18 11:31:21
What is DAMNACANTHAL?
Description
Damnacanthal (477-84-9) is a potent inhibitor of p56lck?tyrosine kinase activity (IC50?= 17 nM for inhibition of autophosphorylation). Displays 7- to 20-fold selectivity over p59fyn?and p60src?tyrosine kinases and > 40-fold to 100-fold selectivity over other kinases.1?Mobilizes intracellular Ca2+?in dermal fibroblasts.2?Inhibits LIM-kinase, impairing cell migration and invasion.3?A potent inhibitor of angiogenesis.4?Inhibits mast cell activation.5
Description
Damnacanthal, an anthraquinone derivative isolated from the noni plant (Morinda citrifolia), has been touted as a phytochemical whose beneficial effects include HIV protein inhibition. But the lack of scientific proof for these claims caused the FDA to issue warning letters to its purveyors during 2002–2006.
The Uses of DAMNACANTHAL
Damnacanthal-induced anti-inflammation is associated with inhibition of NF-κB activity. Damnacanthal is used in the treatment of inflammatory-related diseases.
What are the applications of Application
Damnacanthal is an immunomodulator and potent inhibitor of p56lck tyrosine kinase
Definition
ChEBI: 3-hydroxy-1-methoxy-9,10-dioxo-2-anthracenecarboxaldehyde is a monohydroxyanthraquinone and an aldehyde.
References
1) Faltynek?et al.?(1995),?Damnacanthal is a highly potent, selective inhibitor of p56lck tyrosine kinase activity; Biochemistry,?34?12404 2) Aoki?et al.?(2000),?Mechanism of damnacanthal-induced ;Ca(2+)](i) elevation in human dermal fibroblasts; Eur. J. Pharmacol.,?387?119 3) Ohashi?et al.?(2014),?Damnacanthal, an effective inhibitor of LIM-kinase, inhibits cell migration and invasion; Mol. Biol. Cell,?25?828 4) Garcia-Vilas?et al.?(2017),?The noni anthraquinone damnacanthal is a multi-kinase inhibitor with potent anti-angiogenic effects; Cancer Letters,?385?1 5) Garcia-Vilas?et al.?(2015),?Damnacanthal inhibits IgE receptor-mediated activation of mast cells; Mol. Immunol.,?65?86
Properties of DAMNACANTHAL
| Melting point: | 218~219℃ |
| Boiling point: | 531.9±50.0 °C(Predicted) |
| Density | 1.461±0.06 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | Soluble in DMSO (up to 7 mg/ml) |
| form | Pale orange solid. |
| pka | 5.26±0.20(Predicted) |
| color | Yellow |
| Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 1 month. |
Safety information for DAMNACANTHAL
Computed Descriptors for DAMNACANTHAL
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran

You may like
-
 Damnacanthal CAS 477-84-9View Details
Damnacanthal CAS 477-84-9View Details
477-84-9 -
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4