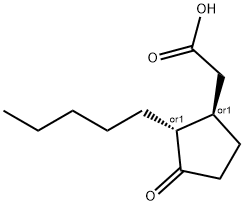
Prohydrojasmon
- CAS NO.:158474-72-7
- Empirical Formula: C15H26O3
- Molecular Weight: 254.37
- MDL number: MFCD08458957
- EINECS: 605-149-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:15:32
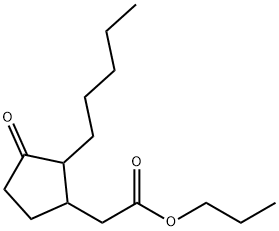
What is Prohydrojasmon ?
Description
Prohydrojasmon (PDJ) [propyl(1RS,2RS)-(3-oxo-2-pentylcyclopentyl) acetate], an analog of Jasmonic acid (JA), was first registered as a plant growth regulator, particularly for coloring apples and grapes. Plant growth regulators are naturally or chemically synthesized substances that play roles in plants' developmental or metabolic processes. Subsequently, similar to JA, PDJ was found to induce direct and indirect defenses against herbivores in non-infested plants[1].
The Uses of Prohydrojasmon
Prohydrojasmon (PDJ), as a plant growth regulator, focuses on fruits, including apples, oranges, mangoes, and grapes. Specifically, it has been applied to enhance mango and red pear fruit coloration. PDJ treatment of crops increases their direct and indirect defenses against several insect pests under open field conditions. A report on PDJ treatment of non-infested maize plants suggests a reduction in the weights and survival rates of the common armyworm, Mythimna separata (Walker), larvae and their increased attraction to Cotesia kariyai Watanabe, a parasitoid wasp of the herbivore. Furthermore, two-spotted spider mites (Tetranychus urticae Koch) laid significantly fewer eggs on PDJ-treated leaf disks than on control leaf disks. PDJ treatment of non-infested lima bean plants induced the production of volatiles, including (E)-β-ocimene and (E)-4,8-dimethyl-1,3,7-nonatriene (DMNT), which attract Phytoseiulus persimilis Athias-Henriot, a predatory mite. Moreover, under greenhouse conditions, PDJ treatment of tomato plants negatively affects the damage caused by thrips[3].
Biological Functions
The activity of Prohydrojasmon may be similar to that of endogenous JA. It has been shown to affect several physiological processes, including senescence, leaf abscission, fruit ripening, coloration, and pigment accumulation. Other studies have shown that PDJ is an elicitor in plants to stimulate the accumulation of secondary metabolites, including phenolics, anthocyanins, terpenoids, and glucosinolates[2].
References
[1] Kengo Yoshida. “Effects of Prohydrojasmon on the Number of Infesting Herbivores and Biomass of Field-Grown Japanese Radish Plants.” Frontiers in Plant Science (2021): 695701.
[2] Shinya Takahashi. “Prohydrojasmon Promotes the Accumulation of Phenolic Compounds in Red Leaf Lettuce.” Plants-Basel 10 9 (2021).
[3] Haidar Rafid Azis. “Effect of Prohydrojasmon on the Growth of Eggplant and Komatsuna.” Plants-Basel (2020).
Properties of Prohydrojasmon
| Boiling point: | 340.2±15.0 °C(Predicted) |
| Density | 0.967±0.06 g/cm3(Predicted) |
| storage temp. | 0-6°C |
| solubility | Chloroform (Slightly), Hexane (Slightly), Methanol (Slightly) |
| form | Oil |
| color | Pale Yellow to Light Yellow |
| InChI | InChI=1S/C15H26O3/c1-3-5-6-7-13-12(8-9-14(13)16)11-15(17)18-10-4-2/h12-13H,3-11H2,1-2H3 |
| EPA Substance Registry System | Cyclopentaneacetic acid, 3-oxo-2-pentyl-, propyl ester (158474-72-7) |
Safety information for Prohydrojasmon
Computed Descriptors for Prohydrojasmon
| InChIKey | IPDFPNNPBMREIF-UHFFFAOYSA-N |
| SMILES | C1(CC(OCCC)=O)CCC(=O)C1CCCCC |
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 4-Hydrazinobenzoic acid 3,4-Dibenzyloxybenzaldehyde Electrolytic Iron Powder 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 4-HYDROXY BENZYL ALCOHOL 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) S-2-CHLORO PROPIONIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 3-(Hydroxymethyl)benzoate N-Boc-2-chloroethylamine 1-Bromo-2-methoxy-3-nitrobenzene N-Methyl-3-cyclopenten-1-amine 2-Bromo-3-hydroxybenzaldehyde 1H-indazole-5-carboxamideRelated products of tetrahydrofuran



You may like
-
 7441-43-2 98%View Details
7441-43-2 98%View Details
7441-43-2 -
 1260741-78-3 6-Bromo-3-iodo-1-methyl-1H-indazole 98%View Details
1260741-78-3 6-Bromo-3-iodo-1-methyl-1H-indazole 98%View Details
1260741-78-3 -
 (3-Benzyloxypropyl)triphenyl phosphonium 98%View Details
(3-Benzyloxypropyl)triphenyl phosphonium 98%View Details
54314-85-1 -
 4-bromo-3,5-dimethylbenzenesulfonyl chloride 1581266-79-6 98%View Details
4-bromo-3,5-dimethylbenzenesulfonyl chloride 1581266-79-6 98%View Details
1581266-79-6 -
 2490430-37-8 98%View Details
2490430-37-8 98%View Details
2490430-37-8 -
 N-(5-Amino-2-methylphenyl)acetamide 5434-30-0 98%View Details
N-(5-Amino-2-methylphenyl)acetamide 5434-30-0 98%View Details
5434-30-0 -
 124371-59-1 98%View Details
124371-59-1 98%View Details
124371-59-1 -
 53857-52-2 98%View Details
53857-52-2 98%View Details
53857-52-2