clorexolone
- CAS NO.:2127-01-7
- Empirical Formula: C14H17ClN2O3S
- Molecular Weight: 328.818
- MDL number: MFCD00867311
- EINECS: 2183426
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-04 17:34:35
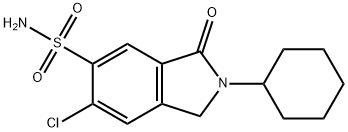
What is clorexolone?
Originator
Speciatensol,Specia,France,1966
The Uses of clorexolone
Chlorexolone is a sulfonamide based antibiotic.
The Uses of clorexolone
Chlorexolone is a low-ceiling sulfonamide diuretic.
Definition
ChEBI: Clorexolone is an organic molecular entity.
Manufacturing Process
4-Chlorophthalimide (263 g) was reacted in amyl alcohol (2.6 l) with
cyclohexylamine (143.5 g, 1 mol) at reflux temperature for 16 hours to give
N-cyclohexyl-4-chlorophthalimide (250 g, 66%) as a solid, MP 134°C to
136°C.
N-Cyclohexyl-1-chlorophthalimide (250 g) was dissolved in glacial acetic acid
(2.5 l), concentrated hydrochloric acid (555 ml) and tin (278 g) were added
and the suspension was heated on a steam bath for 16 hours. The cooled
solution was filtered and concentrated to dryness in vacuo to give a white
solid. This solid was dissolved in water and the precipitated oil extracted with
chloroform. The chloroform solution was dried and concentrated in vacuo to
give a solid which, after recrystallization, yielded 5-chloro-2-
cyclohexylisoindolin-1-one (43%), MP 140°C to 142°C.
5-Chloro-2-cyclohexylisoindolin-1-one (102.9 g) was dissolved in concentrated
sulfuric acid (665 ml); potassium nitrate (723 g) in concentrated sulfuric acid
(166 ml) was added at 0 °C. The reaction mixture was allowed to warm to
room temperature and stirred at 25°C for 12 hours. The reaction mixture was
poured onto ice to give a cream solid which, after recrystallization from
benzene, gave 5-chloro-2-cyclohexyl-6-nitroisoindolin-1-one (46.7 g, 44%) as
a white solid, MP 164°C to 168°C.
5-Chloro-2-cyclohexyl-6-nitroisoindolin-1-one (93.9 g) was reduced in
concentrated hydrochloric acid (1,970 ml) with stannous chloride (376 g). The
reaction temperature rose to 70°C. The resulting solution was cooled in ice
and filtered. The product was washed well with water, filtered and dried to
give 6-amino-5-chloro-2-cyclohexylisoindolin-1-one (74.1 g, 87.6%) which,
after recrystallization from benzene, had a MP of 216°C to 218°C.
6-Amino-5-chloro-2-cyclohexylisoindolin-1-one (42.5 g) was dissolved in
concentrated hydrochloric acid (425 ml) and the solution diazotized by the
addition of sodium nitrite (21.25 g) in water (125 ml). The resulting
diazonium salt solution was added to a solution of liquid sulfur dioxide (93 ml)
in glacial acetic acid (243 ml) containing cuprous chloride (2.25 g). A yellow
solid was precipitated; this was filtered off, washed, dried and recrystallized
from benzene to give 5-chloro-2-cyclohexylisoindolin-1-one-6-sulfonyl chloride
(45 g, 80%) as a cream solid, MP 171°C to 174°C.
This sulfonyl chloride (23.7 g) was reacted with liquid ammonia (237 ml) to
give 5-chloro-2-cyclohexyl-6-sulfamoylisoindolin-1-one (14.2 g, 53%). MP
259°C to 261°C.
Therapeutic Function
Diuretic
Properties of clorexolone
| Melting point: | 266-268° |
| storage temp. | Refrigerator |
| solubility | DMSO (Slightly, Heated), Methanol (Very Slightly, Heated) |
| form | Solid |
| color | White to Off-White |
Safety information for clorexolone
Computed Descriptors for clorexolone
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
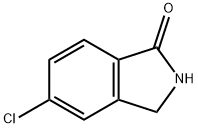

You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1