Calusterone
- CAS NO.:17021-26-0
- Empirical Formula: C21H32O2
- Molecular Weight: 316.48
- MDL number: MFCD00866255
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-07-12 16:37:11
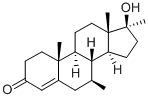
What is Calusterone?
Originator
Methosarb,Upjohn,US,1973
The Uses of Calusterone
Synthetic androgen epimeric with Bolasterone (B674970). Has been used in treatment of breast cancer. Controlled substance (anabolic steroid).
Definition
ChEBI: Calusterone is a 3-hydroxy steroid. It has a role as an androgen.
Manufacturing Process
As described in US Patent 3,029,263, one possibility is a multistep synthesis
starting from 3β,17β-dihydroxy-17α-methyl-5-androstene.
Alternatively, as described in US Patent 3,341,557, 6-dehydro-17-
methyltestosterone may be used as the starting material. A mixture of 0.4 g
of cuprous chloride, 20 ml of 4M methylmagnesium bromide in ether and 60
ml of redistilled tetrahydrofuran was stirred and cooled in an ice bath during
the addition of a mixture of 2.0 g of 6-dehydro-17-methyltestosterone, 60 ml
of redistilled tetrahydrofuran and 0.2 g of cuprous chloride. The ice bath was
removed and stirring was continued for four hours. Ice and water were then
carefully added, the solution acidified with 3 N hydrochloric acid and extracted
several times with ether. The combined ether extracts were washed with a
brine-sodium carbonate solution, brine and then dried over anhydrous
magnesium sulfate, filtered and then poured over a 75-g column of
magnesium silicate (Florisil) packed wet with hexanes (Skellysolve B). The
column was eluted with 250 ml of hexanes, 0.5 liter of 2% acetone, two liters
of 4% acetone and 3.5 liters of 6% acetone in hexanes.
Four 250-ml fractions were collected followed by 150 ml fractions. The
residues from fractions 8 to 16 were combined and rechromatographed over a
125-g column of magnesium silicate. The solumn was eluted with 6% acetone
in hexanes which was collected in 150 ml portions. Fractions 18 to 29 were
combined and dissolved in acetone, decolorized with charcoal, and
recrystallized from acetone. One gram of a crystalline mixture of the 7epimers
of 7,17-dimethyltestosterone was obtained melting at 120° to 140°C.
Therapeutic Function
Antineoplastic
Properties of Calusterone
| Melting point: | 127-129° |
| Boiling point: | 463.29°C (rough estimate) |
| alpha | D +57° (CHCl3) |
| Density | 0.9575 (rough estimate) |
| refractive index | 1.5100 (estimate) |
| solubility | Chloroform (Slightly), Ethyl Acetate (Slightly) |
| form | Solid |
| pka | 15.13±0.70(Predicted) |
| color | White to Off-White |
Safety information for Calusterone
Computed Descriptors for Calusterone
New Products
4-Fluorophenylacetic acid 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate (6-METHYL-[1,3]DITHIOLO[4,5-b]QUINOXALIN-2-ONE INDAZOLE-3-CARBOXYLIC ACID 4-IODO BENZOIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1