BKM120 (NVP-BKM120, Buparlisib)
- CAS NO.:944396-07-0
- Empirical Formula: C18H21F3N6O2
- Molecular Weight: 410.39
- MDL number: MFCD18251596
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-23 21:30:31
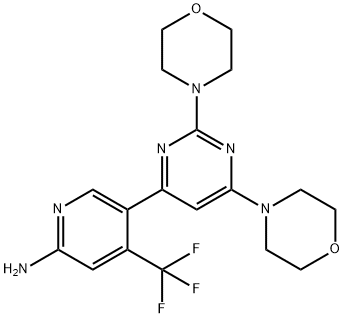
What is BKM120 (NVP-BKM120, Buparlisib)?
Description
Buparlisib (944396-07-0) is potent pan-Class I PI3-kinase inhibitor (IC50’s: p110α = 52 nM, p110? = 166 nM, p110δ = 116 nM. P110g = 262 nM).1,2 It inhibited microtubule dynamics at in vitro concentrations >1μM and doses above 50mg/kg in mice.3 Buparlisib lead to a precipitous drop in DNA synthesis in a mouse model of BRCA1-linked triple-negative breast cancer with less affect in normal tissue.4
The Uses of BKM120 (NVP-BKM120, Buparlisib)
A selective Class I PI3K inhibitor of p110α, p110β, p110δ and p110γ with IC50s of 50-300 nM.
The Uses of BKM120 (NVP-BKM120, Buparlisib)
NVP-BKM 120 is a novel anti-tumor active compound that is selective in that it inhibits specifically PI3 kinase activating cell death in glioma cells. Glioma cells being those that proliferate from tumors in the brain or the spine.
What are the applications of Application
BKM120 is BKM120 is a PI 3-kinase family inhibitor
Definition
ChEBI: BKM120 is an aminopyridine that is 4-(trifluoromethyl)pyridin-2-amine substituted at position 5 by a 2,6-di(morpholin-4-yl)pyrimidin-4-y group. A selective PI3K inhibitor with anti-tumour properties. It has a role as an EC 2.7.1.137 (phosphatidylinositol 3-kinase) inhibitor and an antineoplastic agent. It is a member of morpholines, an aminopyrimidine, an aminopyridine and an organofluorine compound.
References
Burger et al. (2011), Identification of NVP-BKM120 as a Potent, Selective, Orally Bioavailable Class I PI3 Kinase Inhibitor for Treating Cancer; ACS Med. Chem. Lett. 2 774 Maira et al. (2012), Identification and Characterization of NVP-BKM120, an Orally Available Pan-Class I PI3-Kinase Inhibitor; Mol. Cancer Ther. 11 317 Brachmann et al. (2012), Characterization of the mechanism of action of the pan class I PI3K inhibitor NVP-BKM120 across a broad range of concentrations; Mol. Cancer Ther. 11 1747 Juvekar et al. (2016), Phosphoinositide 3-kinase inhibitors induce DNA damage through nucleoside depletion; Proc. Natl. Acad. Sci USA. 113 E4338
Properties of BKM120 (NVP-BKM120, Buparlisib)
| Melting point: | 143-147°C |
| Boiling point: | 645.7±65.0 °C(Predicted) |
| Density | 1.382 |
| storage temp. | -20°C |
| solubility | Soluble in DMSO (15 mg/ml) |
| form | White powder. |
| pka | 5.94±0.50(Predicted) |
| color | White |
| Stability: | Stable for 1 year as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
Safety information for BKM120 (NVP-BKM120, Buparlisib)
Computed Descriptors for BKM120 (NVP-BKM120, Buparlisib)
New Products
Indole Methyl Resin tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Boc-His(Boc)-OH 2-CTC Resin 4-Chloro-7-tosy1-7Hpyrrolo[2,3-d]pyrimidine 5,7-Dibromo-1H-indole 2,5-dichloro-N-hydroxy-4,6-dimethylpyridine-3-carboximidamide 2,2-Dimethoxy-7-azaspiro[3.5]nonane hydrochloride 4-chloromethyl-5-methyl-1,3-dioxol-2-one (DMDO-Cl) R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDIIMIDAZOLE 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID 4-((2-hydroxyethyl)thio)benzoic acid 1-(TERT-BUTOXYCARBONYL)-2-PYRROLIDINONE Methyl 6-methylnicotinate 3-Pyridineacrylic acid tert-Butyl carbazate TETRAHYDRO-2H-PYRAN-3-OL 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile 2,4-dihydroxybenzaldehyde 1,3-Diethyl-1,3-Diphenylurea Methyl 2-methylquinoline-6-carboxylateRelated products of tetrahydrofuran
You may like
-
 Pyridine 99.5% HPLC /UV SpectroscopyView Details
Pyridine 99.5% HPLC /UV SpectroscopyView Details
110-86-1 -
 Guanine , 99%View Details
Guanine , 99%View Details
73-40-5 -
 Piperazine Spot supply, best priceView Details
Piperazine Spot supply, best priceView Details
110-85-0 -
 Potassium Hydroxide 90%View Details
Potassium Hydroxide 90%View Details
1310-58-3 -
 Dibutyl PhthalateView Details
Dibutyl PhthalateView Details
84-74-2 -
 Imidazole Spot supply, competitive priceView Details
Imidazole Spot supply, competitive priceView Details
288-32-4 -
 Octadecyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate 98% (GC)View Details
Octadecyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate 98% (GC)View Details
2082-79-3 -
 Thiourea 99% ARView Details
Thiourea 99% ARView Details
62-56-6
