Azilsartan kaMedoxoMil
- CAS NO.:863031-24-7
- Empirical Formula: C30H25KN4O8
- Molecular Weight: 608.65
- MDL number: MFCD24849277
- EINECS: 1308068-626-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-10-29 15:43:00
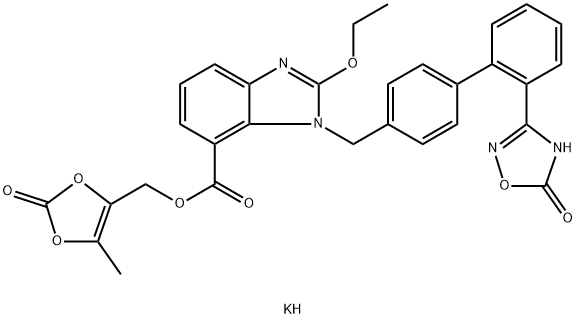
What is Azilsartan kaMedoxoMil?
The Uses of Azilsartan kaMedoxoMil
Azilsartan Kamedoxomil is an angiotensin II receptor blocker.
Definition
ChEBI: An organic potassium salt that is the monopotassium salt of azilsartan medoxomil. A prodrug for azilsartan, it is used for treatment of hypertension.
Clinical Use
Azilsartan kamedoxomil, developed by Takeda Pharmaceuticals, was approved for the treatment of hypertension and launched in the U.S. under the brand name Edarbi. Edarbi is a prodrug that undergoes rapid hydrolysis to liberate azilsartan, the active ingredient (TAK-536, 39). As the 8th angiotensin receptor blocker (ARB) to enter the world market, azilsartan kamedoxomil can function as monotherapy or in combination with other antihypertensive agents. In several clinical studies, monotherapeutic azilsartan kamedoxomil showed superior antihypertensive activity and a favorable safety/tolerability profile in patients compared with other established therapeutics, including valsartan, olmesartan medoxomil, candesartan, and telmisartan.In late 2011,Takeda announced that FDA also approved the fixed-dose combination tablet of azilsartan kamedoxomil with chlorthalidone under the trade name of Edarbyclor.
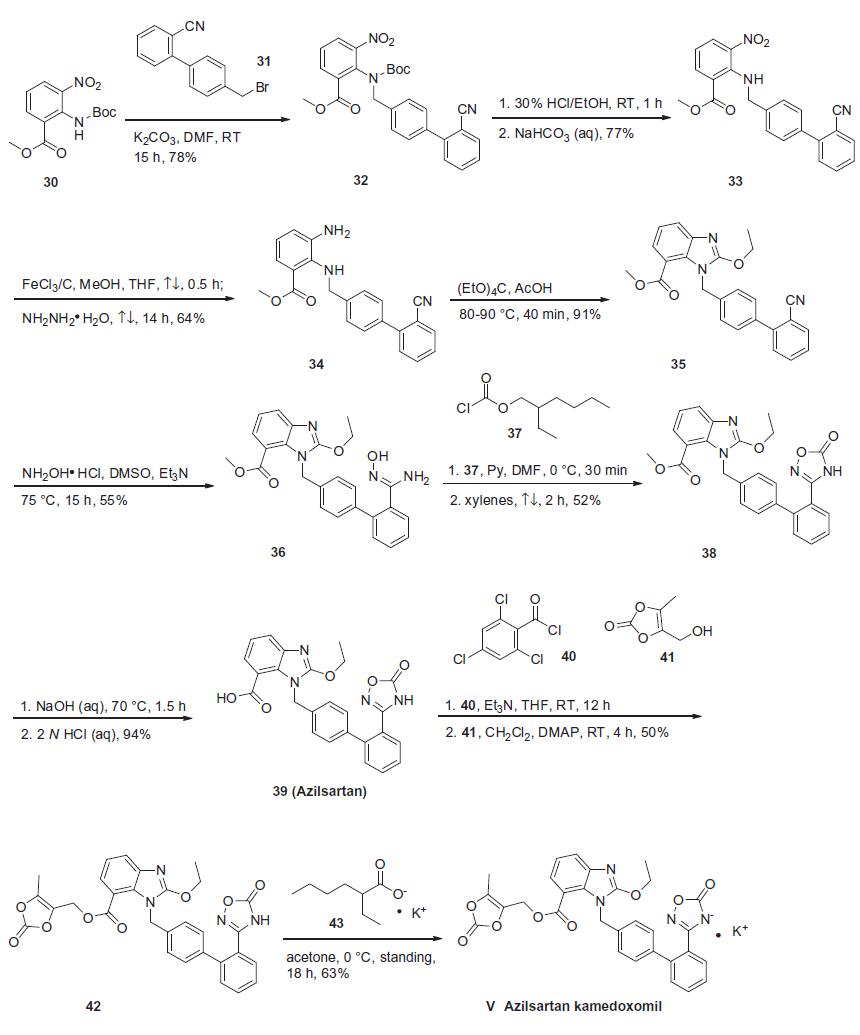
Synthesis
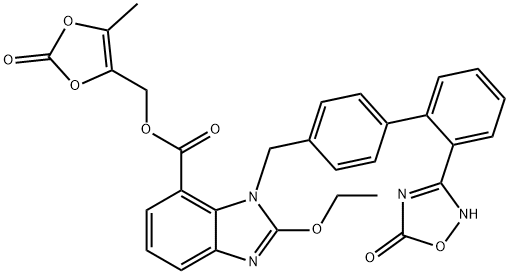
Based on the synthesis of azilsartan, the process-scale approach to azilsartan kamedoxomil is described in the scheme. The synthesis started with commercially available methyl 2-[(tert-butoxycarbonyl)amino]-3-nitrobenzoate (30), which can also be prepared by several different routes.41,42 Alkylation of 30 with diaryl bromide 31 gave benzylamine 32 in 78% yield, which was followed by deprotection with 30% ethanolic HCl and alkalinization to produce amine 33 in 77% yield. The nitro group within 33 was reduced with hydrazine hydrate and a catalytic amount of ferric chloride to afford 2,3-diaminobenzoate 34 in 64% yield. Ring formation was achieved by treatment of 34 with tetraethoxymethane and acetic acid to produce benzimidazole 35 in 91% yield.37 The addition of hydroxylamine to the cyano group of 35 provided amidoxime 36 in 55% yield, which underwent immediate cyclization upon treatment with 2-ethylhexyl chloroformate 37 in refluxing xylenes to give oxadiazolone 38 in 52% yield. Hydrolysis of 38 gave azilsartan (39, TAK-536) in 94% yield. In the presence of perchlorobenzoyl chloride 40 and triethylamine, carboxylic acid 39 was converted to the mixed acid anhydride intermediate, which when condensed with alcohol 41 furnished benzoate 42 in 50% yield. Salt preparation of 42 was accomplished with potassium 2-ethylhexyl carboxylate 43 affording azilsartan kamedoxomil (V) in 63% yield.
Properties of Azilsartan kaMedoxoMil
| Melting point: | 193 - 195°C |
| storage temp. | Refrigerator, Under inert atmosphere |
| solubility | DMSO (Slightly), Methanol (Slightly) |
| form | Solid |
| color | White to Off-White |
Safety information for Azilsartan kaMedoxoMil
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Azilsartan kaMedoxoMil
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
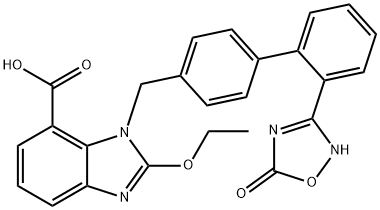
![(Z)-Methyl 2-ethoxy-3-((2'-(N'-hydroxycarbaMiMidoyl)biphenyl-4-yl)Methyl)-3H-benzo[d] iMidazole-4-carboxylate](https://img.chemicalbook.in/CAS/GIF/147403-65-4.gif)
![1H-BenziMidazole-7-carboxylic acid, 1-[[2'-(2,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl)[1,1'-biphenyl]-4-yl]Methyl] -2-ethoxy-, Methyl ester](https://img.chemicalbook.in/CAS/GIF/147403-52-9.gif)
You may like
-
 Azilsartan kamedoxomil 98%View Details
Azilsartan kamedoxomil 98%View Details -
 863031-24-7 98%View Details
863031-24-7 98%View Details
863031-24-7 -
 Azilsartan kamedoxomil 863031-24-7 99%View Details
Azilsartan kamedoxomil 863031-24-7 99%View Details
863031-24-7 -
 863031-24-7 Azilsartan kamedoxomil 98%View Details
863031-24-7 Azilsartan kamedoxomil 98%View Details
863031-24-7 -
 863031-24-7 98%View Details
863031-24-7 98%View Details
863031-24-7 -
 Azilsartan kamedoxomil 99%View Details
Azilsartan kamedoxomil 99%View Details
863031-24-7 -
 863031-24-7 Azilsartan kamedoxomil 98%View Details
863031-24-7 Azilsartan kamedoxomil 98%View Details
863031-24-7 -
 AZILSARTAN KAMEDOXOMIL IH 95-99 %View Details
AZILSARTAN KAMEDOXOMIL IH 95-99 %View Details
863031-24-7
