Amafolone
- CAS NO.:50588-47-1
- Empirical Formula: C19H31NO2
- Molecular Weight: 305.459
- MDL number: MFCD00864749
- SAFETY DATA SHEET (SDS)
- Update Date: 2022-12-21 16:56:50
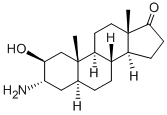
What is Amafolone?
Originator
Amafolone hydrochloride,ZYF Pharm Chemical
Manufacturing Process
A solution of 70% perchloric acid (60 ml) in water (280 ml) was added to a
stirred solution of 5α-androst-2-en-17-one (200.0 g) in ether (1.15 l) at 15°C
followed by portionwise addition of N-bromacetamide (112.0 g) over 10 min.
After stirring for 1 h the precipitated white crystalline solid was filtered off,
washed neutral with ether and water and crystallised to give 2β-hydroxy-3α-
bromo-5α-androstan-17-one (172.0 g). The ether layer of the filtrate was
washed neutral, dried (Na2SO4) and concentrated to give a second crop (28.0
g). The two crops (200.0 g) were suspended in methanol (1 L), 10 N aqueous
potassium hydroxide solution (100 ml) added and the mixture slowly distilled
over 45 min. Addition of water precipitated the product as a white solid which was filtered off, washed with water, dried and 88.0 g of 2β,3β-epoxy-5α-
androstan-17-one were obtained (crystallisation from ether:light petrol).
A solution of the 2β,3β-epoxy-5α-androstan-17-one (30.0 g) in ethanol (130
ml), water (15 ml) and liquid ammonia was heated in an autoclave at 150°C
for 6 h and the resultant crystalline suspension evaporated to dryness. Water
(35 ml) and acetic acid (36 ml) were added and the solution kept at 90°C for
1 h, cooled and excess water added. The precipitated material was filtered off
and the filtrate made alkaline with aqueous 10 N potassium hydroxide solution
to precipitate a white solid which was filtered off, washed neutral with water,
dissolved in methylene chloride, the solution dried (Na2SO4) concentrated and
ether added to give 2β-hydroxy-3α-amino-5α-androstan-17-one (14.3 g),
melting point l92°-195°C.
In practice it is usually used as hydrochloride.
Therapeutic Function
Antiarrhythmic
Safety information for Amafolone
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran
You may like
-
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 88491-46-7 98%View Details
88491-46-7 98%View Details
88491-46-7 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4