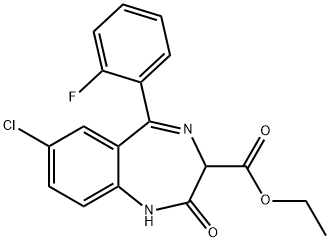
7-chloro-2,3-dihydro-2,2-dihydroxy-5-phenyl-1H-1,4-benzodiazepine-3-carboxylic acid
- CAS NO.:23887-31-2
- Empirical Formula: C16H11ClN2O3
- Molecular Weight: 314.72
- EINECS: 245-926-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2022-12-21 16:56:50
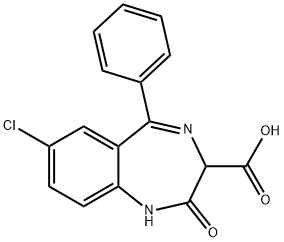
What is 7-chloro-2,3-dihydro-2,2-dihydroxy-5-phenyl-1H-1,4-benzodiazepine-3-carboxylic acid?
Absorption
Clorazepate is a prodrug for nordiazepam, and this conversion occurs almost entirely before entering systemic circulation. Therefore, the pharmacokinetic parameters of clorazepate are based on nordiazepam concentrations. In healthy volunteers given a 20 mg oral dose of clorazepate, nordiazepam had a peak plasma level of 356 ng/mL, which was reached approximately 0.9 h after the administration of clorazepate. The plasma levels of nordiazepam increase proportionally with the clorazepate dose and show moderate accumulation with repeated administration. The oral bioavailability of clorazepate is 91%.
Toxicity
The overdosage of benzodiazepines, such as clorazepate, is characterized by central nervous system (CNS) depression ranging from drowsiness to coma. In mild to moderate cases, symptoms can include drowsiness, confusion, dysarthria, lethargy, hypnotic state, diminished reflexes, ataxia, and hypotonia. Paradoxical or disinhibitory reactions are rare but may occur. In severe cases, patients experiencing a benzodiazepine overdose may develop respiratory depression and coma. Overdosage of benzodiazepines in combination with other CNS depressants may be fatal. General supportive measures, including intravenous fluids and airway management, should be used to manage a benzodiazepine overdose. The use of flumazenil for the complete or partial reversal of benzodiazepine-sedative effects during an overdose can lead to withdrawal and adverse reactions. Refer to the product label of clorazepate for additional overdosage management recommendations.
In rats, the oral LD50 of clorazepate is 1320 mg/kg, and in monkeys, it exceeds 1600 mg/kg. In animal reproduction studies, clorazepate did not affect fertility indices or the reproductive capacity of adult animals.
Background
Clorazepic acid (clorazepate) is a water-soluble benzodiazepine with muscle-relaxant and anticonvulsant actions effective in the treatment of anxiety. Following administration, clorazepate is rapidly converted to nordiazepam (N-desmethyldiazepam), its active metabolite, before entering systemic circulation. Similar to other benzodiazepines, the active metabolite of clorazepate enhances the binding of gamma-aminobutyric acid (GABA) to the GABA type A (GABA-A) receptor, which promotes channel opening and neuronal hyperpolarization. The concomitant use of clorazepate and opioids may result in profound sedation, respiratory depression, coma, and death. Also, the use of clorazepate exposes users to users to the risks of abuse, misuse, and addiction, and its continued use may lead to significant physical dependence. In September 2020, a black box warning describing these risks was included on the product label of benzodiazepines as per FDA regulation. Clorazepate and its active metabolite, nordiazepam, are present in breast milk.
Indications
Clorazepate is indicated for the management of anxiety disorders or the short-term relief of the symptoms of anxiety. It is also used as adjunctive therapy in the management of partial seizures and for the symptomatic relief of acute alcohol withdrawal.
Definition
ChEBI: A 1,4-benzodiazepinone in which the oxo group is at position 2, and which is substituted at positions 3, 5, and 7 by carboxy, phenyl and chloro groups, respectively.
Pharmacokinetics
Pharmacologically, clorazepate has the characteristics of benzodiazepines. Studies in healthy men have shown that clorazepate has depressant effects on the central nervous system. Since orally administered clorazepate dipotassium is rapidly decarboxylated to form nordiazepam, there is essentially no circulating parent drug. The use of benzodiazepines, including clorazepate, exposes users to abuse, misuse, and addiction risks, which can lead to overdose and death. Patients treated with clorazepate may also develop suicidal behavior and ideation, and the use of clorazepate may cause interference with psychomotor performance. The concomitant use of clorazepate with opioids may result in profound sedation, respiratory depression, coma, and death.
Metabolism
Clorazepate is metabolized in the liver and excreted mainly through urine. Following oral administration, clorazepate is decarboxylated by the gastric acid of the stomach before absorption. The primary metabolite, nordiazepam, is further metabolized by hydroxylation. The major urinary metabolite is conjugated oxazepam (3-hydroxynordiazepam), and smaller amounts of conjugated p-hydroxynordiazepam and nordiazepam are also found in the urine.
Properties of 7-chloro-2,3-dihydro-2,2-dihydroxy-5-phenyl-1H-1,4-benzodiazepine-3-carboxylic acid
| Boiling point: | 563.9±50.0 °C(Predicted) |
| Density | 1.46±0.1 g/cm3(Predicted) |
| pka | 0.80±0.40(Predicted) |
Safety information for 7-chloro-2,3-dihydro-2,2-dihydroxy-5-phenyl-1H-1,4-benzodiazepine-3-carboxylic acid
Computed Descriptors for 7-chloro-2,3-dihydro-2,2-dihydroxy-5-phenyl-1H-1,4-benzodiazepine-3-carboxylic acid
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran
![3-[(4-chlorophenyl)amino]propanoic acid](https://img.chemicalbook.in/CAS/GIF/21617-19-6.gif)


You may like
-
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 88491-46-7 98%View Details
88491-46-7 98%View Details
88491-46-7 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4