SAFETY INFORMATION
| Signal word | Warning |
|---|---|
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
COMPUTED DESCRIPTORS
| Molecular Weight | 254.32 g/mol |
|---|---|
| XLogP3 | 4.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 3 |
| Exact Mass | 254.130679813 g/mol |
| Monoisotopic Mass | 254.130679813 g/mol |
| Topological Polar Surface Area | 40.5 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 280 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
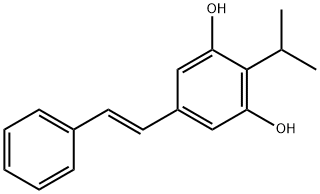
description
Tapinarof is a novel, first-in-class, small-molecule AhR agonist that is indicated for the treatment of adult psoriasis. It is available as a topical cream to be applied to the affected area once daily. Tapiranof was first discovered as a metabolite (3,5-dihydroxy-4-isopropylstilbene) produced in Photorhabdus luminescens, a gram-negative bacillus that lives symbiotically with the Heterorhabditis nematodes. In 1959, it was noticed that Heterorhabditis with a high amount of 3,5-dihydroxy-4-isopropylstilbene did not putrefy once dead, thus suggesting its potential anti-inflammatory activity. Tapinarof received initial approval from the FDA in 2022.