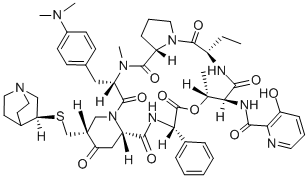
Virginiamycin S1
Synonym(s):Antibiotic 899;Staphylomycin S
- CAS NO.:23152-29-6
- Empirical Formula: C43H49N7O10
- Molecular Weight: 823.89
- MDL number: MFCD00864857
- EINECS: 245-462-6
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-10-28 23:16:16
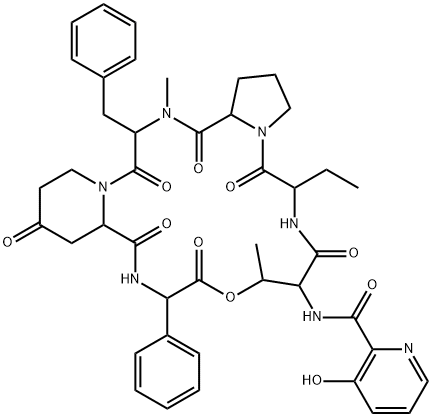
What is Virginiamycin S1?
The Uses of Virginiamycin S1
Virginiamycin complex is defined as a mixture of 75% ostreogrycin A (virginamycin M1) and 25% virginiamycin S1, together with the less abundant S analogues. As the two major components have quite different solubilities, these proportions are not readily achieved or used. BioAustralis has isolated and re-combined the individual components to provide the defined components of virginiamycin complex. The composition of the complex is important as Virginiamycin S1 acts a synergist, binding to the conformational change of the peptidyl transferase centre of the 50S ribosome induced by ostreogrycin A.
The Uses of Virginiamycin S1
Virginiamycin S1 is one of a family of depsipeptide antibiotics co-produced with ostreogrycin A and used as a synergistic mixture. Virginiamycin S1 acts a synergist, binding to the conformational change of the peptidyl transferase centre of the 50S ribosome induced by ostreogrycin A.
The Uses of Virginiamycin S1
Virginiamycin S1 is a main component of virginiamycin preparations.
What are the applications of Application
Virginiamycin S1 is a main component of virginiamycin preparations
Biological Activity
virginiamycin s1 is a macrolide antibiotic that reversibly inhibits protein synthesis [1][2][3].virginiamycin complex contains two antibiotics, virginiamycin m1 and virginiamycin s1. streptogramins are divided into class a and class b based on their structures. virginiamycin s1 is a member of the streptogramin b group of antibiotics, which bind the peptide exit tunnel to inhibit the elongation stage of translation. they show good bactericidal activity against methicillin-resistant s. aureus (mrsa), although resistance in mrsa is conferred by the cfr gene. virginiamycin m1 has activity against gram-positive and in select cases gram-negative bacteria. combination of group a and b streptogramins exhibit bactericidal activity [1]. virginiamycin s1 acted synergistically with virginiamycin m1 to irreversibly inhibit protein synthesis in bacteria. in cell-free systems, virginiamycin m1 and virginiamycin s1 bound to the large ribosomal subunit, and the affinity of ribosomes for vs is increased by vm [2][3].
Contact allergens
Like the other streptogramin, pristinamycin, virginiamycin is made of two subunits, virginiamycin S1 and virginiamycin M1. Dermatitis was quite common in people using the formerly available topical virginiamycin. Occupational dermatitis was observed in the pharmaceutical industry, in breeders, and in a surgeon who used topical virginiamycin on postoperative wounds (personal observation).
References
[1]. fair rj, tor y. antibiotics and bacterial resistance in the 21st century. perspect medicin chem. 2014 aug 28;6:25-64.
[2]. kehrenberg c, cuny c, strommenger b, et al. methicillin-resistant and -susceptible staphylococcus aureus strains of clonal lineages st398 and st9 from swine carry the multidrug resistance gene cfr. antimicrob agents chemother. 2009 feb;53(2):779-81.
[3]. parfait r, cocito c. lasting damage to bacterial ribosomes by reversibly bound virginiamycin m. proc natl acad sci u s a. 1980 sep;77(9):5492-6.
Properties of Virginiamycin S1
| Melting point: | 240-242° |
| alpha | D20 -28° (c = 1 in ethanol) |
| storage temp. | -20°C |
| solubility | methanol: soluble5mg/mL |
| form | powder |
| color | white |
Safety information for Virginiamycin S1
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for Virginiamycin S1
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 4-Hydrazinobenzoic acid 3,4-Dibenzyloxybenzaldehyde 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) 4-HYDROXY BENZYL ALCOHOL 3-NITRO-2-METHYL ANILINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 4-methoxy-3,5-dinitropyridine 2-(Cyanocyclohexyl)acetic acid 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride tert-butyl 4- (ureidomethyl)benzylcarbamate diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
![ETHYL[(1-PROPYLPIPERIDIN-2-YL)METHYL]AMINE](https://img.chemicalbook.in/CAS/GIF/901585-97-5.gif)
You may like
-
 Virginiamycin S1 CAS 23152-29-6View Details
Virginiamycin S1 CAS 23152-29-6View Details
23152-29-6 -
 55441-95-7 99%View Details
55441-95-7 99%View Details
55441-95-7 -
 N-Vinylformamide 99%View Details
N-Vinylformamide 99%View Details
13162-05-5 -
 Chloro Uracil 1820-81-1 99%View Details
Chloro Uracil 1820-81-1 99%View Details
1820-81-1 -
 207557-35-5 99%View Details
207557-35-5 99%View Details
207557-35-5 -
 2-ethyl-6-methyl-3-hydroxypyridine succinate 99%View Details
2-ethyl-6-methyl-3-hydroxypyridine succinate 99%View Details
127464-43-1 -
 2-ETHYLPYRIDINE 100-71-0 99%View Details
2-ETHYLPYRIDINE 100-71-0 99%View Details
100-71-0 -
 181228-33-1 (S)-Methyl 3-amino-2-((tert-butoxycarbonyl)amino)propanote Hydrochloride (DAP-OMe. HCl) 99%View Details
181228-33-1 (S)-Methyl 3-amino-2-((tert-butoxycarbonyl)amino)propanote Hydrochloride (DAP-OMe. HCl) 99%View Details
181228-33-1