URANIUM(VI) OXIDE
- CAS NO.:1344-58-7
- Empirical Formula: O3U
- Molecular Weight: 286.03
- MDL number: MFCD00036303
- EINECS: 215-701-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-08-28 13:53:25
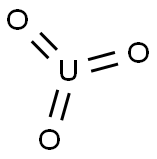
What is URANIUM(VI) OXIDE?
Chemical properties
-100 mesh; has six forms: α is hexagonal brown, β is orange monoclinic, γ is bright yellow rhomb, δ is red cub, ε is brick red tricl, ηis rhomb; UO3 can be obtained by thermal decomposition of uranyl compounds, e.g, carbonates, oxalates nitrates [KIR83] [CER91]
Chemical properties
Uranium trioxide has been isolated in six well-defined stoichiometric modifications as well as a hypostoichiometric modification, UO2. Similar to U3O8, the trioxide decomposes into lower oxides prior to melting or subliming. Even though UO3 is formally U(VI), a small temperature-dependent paramagnetism exists with molar magnetic susceptibility values ranging from 128 to 157×10?6 cm3/mol. A general trend has been observed for the densities of uranium oxides; an inverse proportionality between the O/U ratio and the density of the material. The trioxide does not deviate from this trend with density values ranging from 6.99 to 8.54 g/cm3, depending on the modification.
The Uses of URANIUM(VI) OXIDE
Uranium trioxide, UO3, is a versatile solid that has important applications in the nuclear fuel cycle.
Purification Methods
The oxide is dissolved in HClO4 (to give a uranium content of 5%), and the solution is adjusted to pH 2 by addition of dilute ammonia. Dropwise addition of 30% H2O2, with rapid stirring, precipitated U(VI) peroxide, the pH being held constant during the precipitation, by addition of small amounts of the ammonia solution. Then H2O2 is added until further quantities caused no change in pH. After stirring for 1hour, the slurry is filtered through coarse filter paper in a Büchner funnel, washed with 1% H2O2 acidified to pH 2 with HClO4, then heated at 350o for three days in a large platinum dish [Baes J Phys Chem 60 878 1956].
Properties of URANIUM(VI) OXIDE
| Melting point: | °Cd ec.) |
| Density | 7,29 g/cm3 |
| solubility | insoluble in H2O; soluble in acid solutions |
| form | orange-yellow crystals |
| color | orange-yellow crystals, crystalline |
| Water Solubility | insoluble H2O; soluble acids [MER06] |
| EPA Substance Registry System | Uranium trioxide (1344-58-7) |
Safety information for URANIUM(VI) OXIDE
Computed Descriptors for URANIUM(VI) OXIDE
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran



You may like
-
 Uranyl oxide, orange CAS 1344-58-7View Details
Uranyl oxide, orange CAS 1344-58-7View Details
1344-58-7 -
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4