Unii-na83F1sjsr
- CAS NO.:706782-28-7
- Empirical Formula: C29H40FN3O8
- Molecular Weight: 577.65
- MDL number: MFCD09970919
- EINECS: 1806241-263-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-17 16:00:36
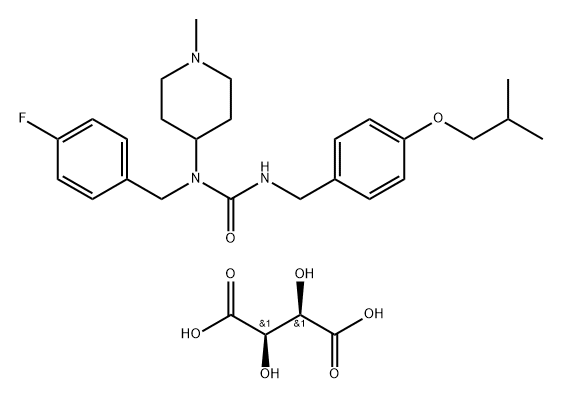
What is Unii-na83F1sjsr?
Description
Pimavanserin, developed by San Diego-based Acadia Pharmaceuticals, is a selective and potent serotonin 2A (5-HT2A) receptor inverse agonist. The USFDA approved this once-daily drug to treat the delusions and hallucinations associated with psychosis as a function of Parkinson’s disease. Pimavanserin has a unique mechanism of action relative to other antipsychotics, behaving as a selective inverse agonist of the serotonin 5-HT2A receptor while exhibiting 40-fold selectivity over the 5-HT2C receptor and having no significant affinity or activity with the 5-HT2B or dopamine receptors.
The Uses of Unii-na83F1sjsr
Pimavanserin tartrate is a 5-HT2A inverse agonist that reverses psychosis-like behaviours and has the potential to treat various other neuropsychiatric disorders such as schizophrenia and Parkinson’s disease.
Definition
ChEBI: A tartrate salt that is the hemitartrate salt of pimavanserin. An atypical antipsychotic that is used for treatment of hallucinations and delusions associated with Parkinson's disease.
Synthesis
Three patent applications filed by Acadia described the process-scale synthetic approach to pimavanserin. The kilogram-scale synthesis began with the alkylation of commercially available 4-hydroxybenzaldehyde (123) with isobutyl bromide (124) under basic conditions. Condensation of the resultant benzaldehyde 125 with hydroxylamine furnished the corresponding oxime 126 in 63% yield from 123. Hydrogenation of 126 catalyzed by Pd/C under acidic conditions produced benzylamine 127, which was isolated as the acetate salt in 41% yield. This compound underwent sodium hydroxide workup followed by reaction with HCl gas and phosgene to deliver isocyanate 128. Nucleophilic attack of this isocyanate by benzylamine 129 (prepared from reductive amination of commercially available N-methylpiperid-4-one 130 with 4-fluorobenzylamine 131) followed by salt formation using tartaric acid in aqueous isopropyl acetate, and THF completed the synthesis of pimavanserin tartrate (XI) in 50% yield over the two-step protocol and a 10.6% overall yield from 123.
Properties of Unii-na83F1sjsr
| storage temp. | Store at -20°C |
| solubility | DMSO:70.0(Max Conc. mg/mL);69.6(Max Conc. mM) |
| form | Solid |
| color | White to off-white |
Safety information for Unii-na83F1sjsr
Computed Descriptors for Unii-na83F1sjsr
Abamectin manufacturer
Honour Lab Limited
Biophore India Pharmaceuticals Pvt Ltd
Archerchem Healthcare Pvt., Ltd. (part of Archerchem Group)
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Mefenamic Acid IP/BP/EP/USP Diclofenac Sodium IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran
![N-[(4-Fluorophenyl)methyl]-1-methyl-4-piperidinamine](https://img.chemicalbook.in/CAS/GIF/359878-47-0.gif)


You may like
-
 706782-28-7 Pimavanserin tartrate 98%View Details
706782-28-7 Pimavanserin tartrate 98%View Details
706782-28-7 -
 706782-28-7 98%View Details
706782-28-7 98%View Details
706782-28-7 -
 Pimavanserin tartrate 98%View Details
Pimavanserin tartrate 98%View Details
706782-28-7 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4