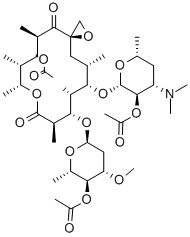
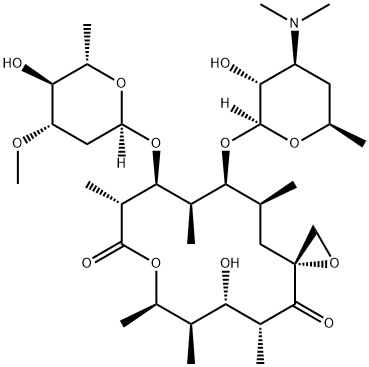
TROLEANDOMYCIN
- CAS NO.:2751-09-9
- Empirical Formula: C41H67NO15
- Molecular Weight: 813.97
- MDL number: MFCD00871198
- EINECS: 220-392-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-10-28 23:16:16
What is TROLEANDOMYCIN?
Toxicity
Troleandomycin inhibits clearance of theophylline and can increase the likelyhood of toxicity in patients recieving theophylline therapy .
Description
Troleandomycin (2751-09-9) is a macrolide antibiotic. Inhibits cytochrome P450 (IC50 = 0-5 μM), thus blocking CYP450-dependent drug oxidation.
The Uses of TROLEANDOMYCIN
Antibacterial.
The Uses of TROLEANDOMYCIN
Troleandomycin, is a macrolide antibiotic, sold in Italy and Turkey. It is a derivative of Oleandomycin (O517500), and acts as a CYP3A4 inhibitor, which may cause drug interactions .
Definition
ChEBI: A semi-synthetic macrolide antibiotic obtained by acetylation of the three free hydroxy groups of oleandomycin. Troleandomycin is only found in individuals that have taken the drug.
Background
A macrolide antibiotic that is similar to erythromycin.
Indications
For the treatment of bacterial infection.
brand name
Tao (Pfizer).
General Description
Triacetyloleandomycin was synthesized by Pfizer Research Laboratories in 1958. It shows a higher and longer-lasting serum level than oleandomycin when administered orally. Triacetyloleandomycin behaves as oleandomycin in vivo, following its hydrolysis by intestinal esterase. However, considerable amounts of the intermediates, the monoacetates and diacetates, are detected in the serum and urine. Prolonged use of triacetyloleandomycin causes damage to the liver as does erythromycin estolate.
Pharmacokinetics
Troleandomycin, like other macrolide antibiotics, inhibits bacterial protein synthesis to prevent growth.
Clinical Use
Oleandomycin contains three hydroxyl groups that aresubject to acylation, one in each of the sugars and one in theoleandolide. The triacetyl derivative retains the in vivo antibacterialactivity of the parent antibiotic but possesses superiorpharmacokinetic properties. It is hydrolyzed in vivo tooleandomycin. Troleandomycin achieves more rapid andhigher plasma concentrations following oral administrationthan oleandomycin phosphate, and it has the additionaladvantage of being practically tasteless. Troleandomycinoccurs as a white, crystalline solid that is nearly insoluble inwater. It is relatively stable in the solid state but undergoeschemical degradation in either aqueous acidic or alkalineconditions.
Because the antibacterial spectrum of activity of oleandomycinis considered inferior to that of erythromycin, thepharmacokinetics of troleandomycin have not been studiedextensively. Oral absorption is apparently good, and detectableblood levels of oleandomycin persist up to 12 hoursafter a 500-mg dose of troleandomycin. Approximately 20%is recovered in the urine, with most excreted in the feces, primarilyas a result of biliary excretion. There is some epigastricdistress following oral administration, with an incidencesimilar to that caused by erythromycin. Troleandomycin isthe most potent inhibitor of cytochrome P450 enzymes of thecommercially available macrolides. It may potentiate the hepatictoxicity of certain anti-inflammatory steroids and oralcontraceptive drugs as well as the toxic effects of theophylline,carbamazepine, and triazolam. Several allergic reactions,including cholestatic hepatitis, have also been reportedwith the use of troleandomycin.
Approved medical indications for troleandomycin arecurrently limited to the treatment of upper respiratory infectionscaused by such organisms as S. pyogenes and S. pneumoniae.It may be considered an alternative to oral forms oferythromycin. It is available in capsules and as a suspension.
Metabolism
Not Available
References
References/Citations 1) Patki et al. (2004), Mechanism-based inhibition of CYP3A4 and CYP3A5 by seven inhibitors; Clin. Pharmacol. Therap., 75 P83 2) Kozlov et al. (2003), Various intracellular compartments cooperate in the release of nitric oxide from glycerol trinitrate in liver; Br. J. Pharmacol., 139 989 3) Ludden et al. (1985), Pharmacokinetic interactions of the macrolide antibiotics; Clin. Pharmacokinet., 10 63
Properties of TROLEANDOMYCIN
| Melting point: | 170 °C |
| Boiling point: | 757°C (rough estimate) |
| alpha | D25 -23° (methanol) |
| Density | 1.1651 (rough estimate) |
| refractive index | 1.6220 (estimate) |
| storage temp. | 0-6°C |
| solubility | Soluble in DMSO (up to 50 mg/ml) or in Ethanol (up to 25 mg/ml). |
| pka | 6.6(at 25℃) |
| form | White solid. |
| color | White |
| Water Solubility | 0.25g/L(28 ºC) |
| Merck | 13,9839 |
| Stability: | Stable for 2 years as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 3 months. |
Safety information for TROLEANDOMYCIN
Computed Descriptors for TROLEANDOMYCIN
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 4-Hydrazinobenzoic acid 3,4-Dibenzyloxybenzaldehyde 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) 4-HYDROXY BENZYL ALCOHOL 3-NITRO-2-METHYL ANILINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 4-methoxy-3,5-dinitropyridine 2-(Cyanocyclohexyl)acetic acid 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride tert-butyl 4- (ureidomethyl)benzylcarbamate diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran



You may like
-
 Troleandomycin CASView Details
Troleandomycin CASView Details -
 55441-95-7 99%View Details
55441-95-7 99%View Details
55441-95-7 -
 N-Vinylformamide 99%View Details
N-Vinylformamide 99%View Details
13162-05-5 -
 Chloro Uracil 1820-81-1 99%View Details
Chloro Uracil 1820-81-1 99%View Details
1820-81-1 -
 207557-35-5 99%View Details
207557-35-5 99%View Details
207557-35-5 -
 2-ethyl-6-methyl-3-hydroxypyridine succinate 99%View Details
2-ethyl-6-methyl-3-hydroxypyridine succinate 99%View Details
127464-43-1 -
 2-ETHYLPYRIDINE 100-71-0 99%View Details
2-ETHYLPYRIDINE 100-71-0 99%View Details
100-71-0 -
 181228-33-1 (S)-Methyl 3-amino-2-((tert-butoxycarbonyl)amino)propanote Hydrochloride (DAP-OMe. HCl) 99%View Details
181228-33-1 (S)-Methyl 3-amino-2-((tert-butoxycarbonyl)amino)propanote Hydrochloride (DAP-OMe. HCl) 99%View Details
181228-33-1