Tirzepatide
- CAS NO.:2023788-19-2
- Empirical Formula: C225H348N48O68
- Molecular Weight: 4813
- EINECS: 200-001-8
- Update Date: 2025-11-20 17:40:16
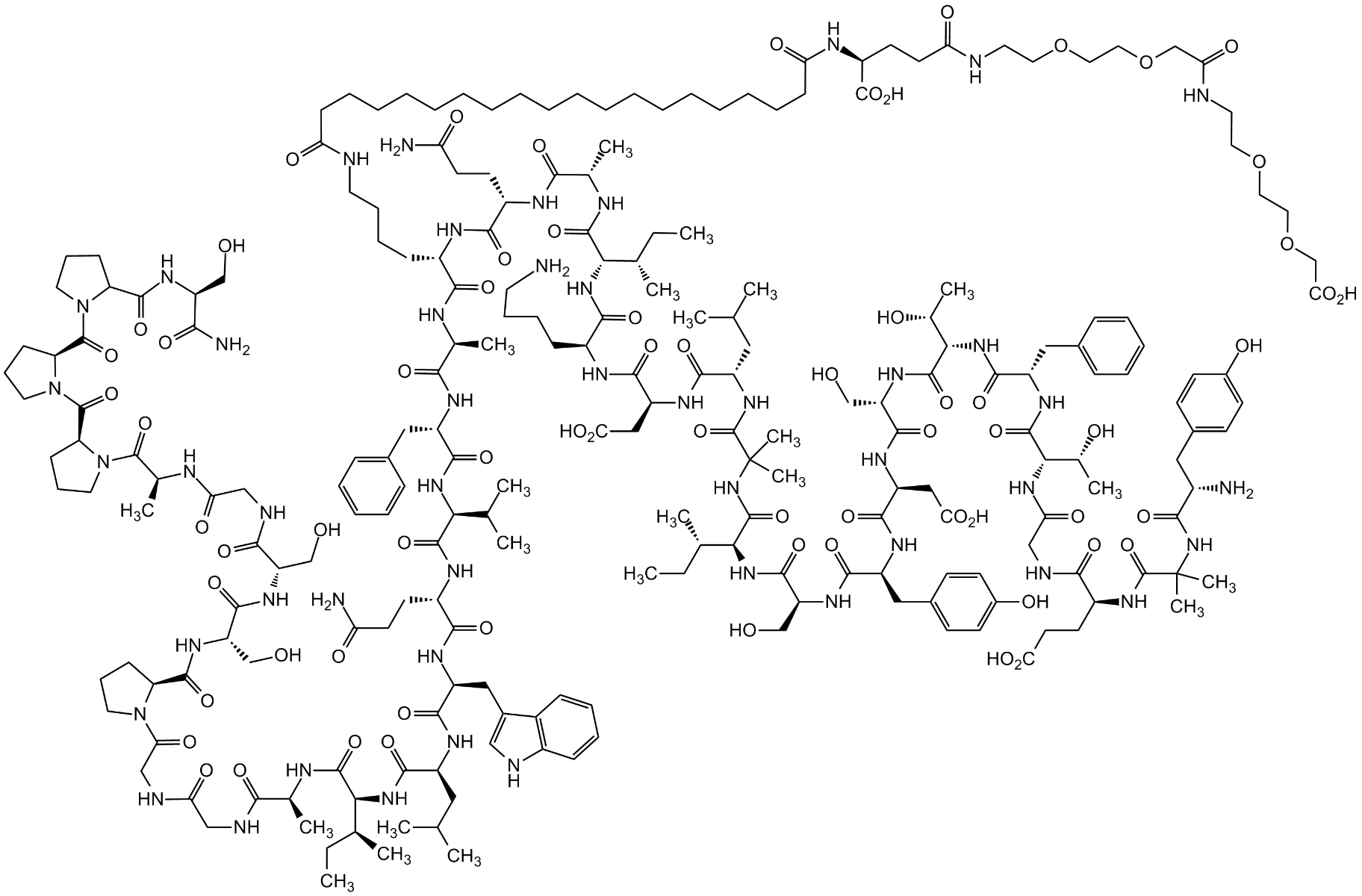
What is Tirzepatide?
Description
Tirzepatide (LY3298176) was developed as a dual agonist to both GLP-1 and gastric inhibitory polypeptide (GIP) receptors (Frias et al., 2018). Similar to GLP-1, GIP is an incretin hormone that functions to induce insulin secretion.
The Uses of Tirzepatide
Tirzepatide is used with a proper diet and exercise program to control high blood sugar in people with type 2 diabetes. Controlling high blood sugar helps prevent kidney damage, blindness, nerve problems, loss of limbs, and sexual function problems.
Definition
Mounjaro® and Ozempic® have distinct active ingredients: tirzepatide and semaglutide, respectively. However, both of these drugs are GLP-1 agonists, which bind to GLP-1 receptors, simulate a feeling of satiety, and signal the pancreas to produce insulin.
Trade name
brand name: Mounjaro
Mechanism of action
It works to stimulate first- and second-phase insulin secretion, and reduces glucagon levels, both in a glucose-dependent manner. Tirzepatide was also shown to delay gastric emptying, lower fasting and postprandial glucose concentration, decrease food intake, 4 and reduce body weight in patients with type 2 diabetes.
Pharmacology
Tirzepatide is a once-weekly GIP (glucose-dependent insulinotropic polypeptide) receptor and GLP-1 (glucagon-like peptide-1) receptor agonist that integrates the actions of both incretins into a single novel molecule. GIP is a hormone that may complement the effects of GLP-1 receptor agonists. In preclinical models, GIP has been shown to decrease food intake and increase energy expenditure therefore resulting in weight reductions, and when combined with GLP-1 receptor agonism, may result in greater effects on markers of metabolic dysregulation such as body weight, glucose and lipids. Tirzepatide is in phase 3 development for adults with obesity or overweight with weight-related comorbidity and is currently under regulatory review as a treatment for adults with type 2 diabetes. It is also being studied as a potential treatment for non-alcoholic steatohepatitis (NASH) and heart failure with preserved ejection fraction (HFpEF). Studies of tirzepatide in obstructive sleep apnea (OSA) and in morbidity/mortality in obesity are planned as well.
Side Effects
The overall safety and tolerability profile of tirzepatide was similar to other incretin-based therapies that have been approved for the treatment of obesity. This said, reported side effects were considerable, especially as dosage levels increased. The most common adverse events were nausea (~30%), diarrhea (~20%), constipation (~15%) and vomiting (~10%).
If tirzepatide gets approved as a both a blood glucose control and anti-obesity agent, it could become a blockbuster drug. However, this isn’t a sure thing. It will have to overcome pricing and reimbursement obstacles, which have plagued similar treatments.
Synthesis
The synthesis process of tirzepatide is as follows: to a reactor was charged dichloromethane (28.6 kg), water (5.4 kg), DTT (4.3 kg), Boc-Fragment 1 + 2 + 3 + 4 (14.3 kg, 8), and TIPS (3.3 kg), resulting in a slurry. The slurry was cooled to less than 10 °C before TFA (162 kg) was added over no more than 1.75 h, resulting in a solution. The solution was warmed to 21 °C and held at this temperature for 3 h. The solution was transferred to a separate reactor, rinsing with TFA (54.3 kg). This solution was cooled to ?10 °C and charged MTBE (125.8 kg) over 2 h. Then, additional MTBE (233 kg) was charged at 17 kg/h, maintaining an internal temperature of less than ?5 °C. The resulting slurry was warmed to 0 °C and then filtered. The wet cake was washed with MTBE (2 × 11 kg/kg relative to 8) and then dried under vacuum at no more than 35 °C to obtain tirzepatide (1, 8.71 kg, 1.81 mol, 81% yield)[1].
Clinical claims and research
Tirzepatide (Eli Lilly), a novel, once-weekly injectable dual glucose-dependent insulinotropic polypeptide (GIP) receptor and GLP-1 RA combination drug, has been developed to treat patients with T2DM. The manufacturer (Eli Lilly) announced the submission of a biologics license application with priority review to the FDA for T2DM on October 27, 2021, with a decision expected in mid-2022.
Mode of action
Tirzepatide has a greater affinity to GIP receptors than to GLP-1 receptors, and this dual agonist behaviour has been shown to produce greater reductions of hyperglycemia compared to a selective GLP-1 receptor agonist. Signaling studies have shown that this is due to tirzepatide mimicking the actions of natural GIP at the GIP receptor. However, at the GLP-1 receptor, tirzepatide shows bias towards cAMP (a messenger associated with regulation of glycogen, sugar and lipid metabolism) generation, rather than β-arrestin recruitment. This combination of preference towards GIP receptor and distinct signaling properties at GLP-1 suggest this biased agonism increases insulin secretion. Tirzepatide has also been shown to increase levels of adiponectin, an adipokine involved in the regulation of both glucose and lipid metabolism, with a maximum increase of 26% from baseline after 26 weeks, at the 10 mg dosage.
Research
Tirzepatide is in phase 3 clinical development at Eli Lilly and Company for blood glucose management in adults with type 2 diabetes, chronic weight management, and obesity-related heart failure with preserved ejection fraction. In addition, Tirzepatide is being studied as a potential treatment for non-alcoholic steatohepatitis (NASH). The molecule comprises a 39 amino acid peptide backbone and a side chain at residue 20. Of the 39 amino acids, 37 are naturally occurring (or coded), while two are noncoded aminoisobutyric acid residues at positions 2 and 13[1].
References
[1] Calley, J. and W. Dhillo. “Effects of the Hormone Kisspeptin on Reproductive Hormone Release in Humans.” 2014. 0.
Properties of Tirzepatide
| storage temp. | Store at -20°C |
| solubility | Soluble in DMSO |
| form | Solid |
| color | White to off-white |
Safety information for Tirzepatide
Computed Descriptors for Tirzepatide
Tirzepatide manufacturer
Sumar biotech LLP
New Products
Indole Methyl Resin tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Boc-His(Boc)-OH 2-CTC Resin 4-Chloro-7-tosy1-7Hpyrrolo[2,3-d]pyrimidine 5,7-Dibromo-1H-indole 2,5-dichloro-N-hydroxy-4,6-dimethylpyridine-3-carboximidamide 2,2-Dimethoxy-7-azaspiro[3.5]nonane hydrochloride 4-chloromethyl-5-methyl-1,3-dioxol-2-one (DMDO-Cl) R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDIIMIDAZOLE 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID 4-((2-hydroxyethyl)thio)benzoic acid 1-(TERT-BUTOXYCARBONYL)-2-PYRROLIDINONE Methyl 6-methylnicotinate 3-Pyridineacrylic acid tert-Butyl carbazate TETRAHYDRO-2H-PYRAN-3-OL 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile 2,4-dihydroxybenzaldehyde 1,3-Diethyl-1,3-Diphenylurea Methyl 2-methylquinoline-6-carboxylateRelated products of tetrahydrofuran





You may like
-
 Mounjaro Tirzepatide 20 Mg InjectionView Details
Mounjaro Tirzepatide 20 Mg InjectionView Details
2023788-19-2 -
 Tirzepatide Usa Delivery, 40 mgView Details
Tirzepatide Usa Delivery, 40 mgView Details
2023788-19-2 -
 Peptides Science Tirzepatide 40 MG Vial, Packaging Type: Bottle, BarsView Details
Peptides Science Tirzepatide 40 MG Vial, Packaging Type: Bottle, BarsView Details
2023788-19-2 -
 Tirzepatide 40 mg PeptideView Details
Tirzepatide 40 mg PeptideView Details
2023788-19-2 -
 Peptide Science Tirzepatide 40 mg Injection, Powder, Packaging Type: BottleView Details
Peptide Science Tirzepatide 40 mg Injection, Powder, Packaging Type: BottleView Details
2023788-19-2 -
 Tirzepatide 40 mg vailView Details
Tirzepatide 40 mg vailView Details
2023788-19-2 -
 Peptide Injection - Tirzepatide 60mg - Packaging Type: Vial, Packaging Size:vial CAS:2023788-19-2View Details
Peptide Injection - Tirzepatide 60mg - Packaging Type: Vial, Packaging Size:vial CAS:2023788-19-2View Details
2023788-19-2 -
 Tirzepatide 60 mg vialView Details
Tirzepatide 60 mg vialView Details
2023788-19-2
