Succinic acid
Synonym(s):Butanedioic acid;Succinic acid
- CAS NO.:110-15-6
- Empirical Formula: C4H6O4
- Molecular Weight: 118.09
- MDL number: MFCD00002789
- EINECS: 203-740-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-26 08:49:36
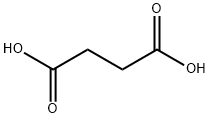
What is Succinic acid?
Toxicity
Oral rat LD50: 2260 mg/kg
Description
Succinic acid is a dicarboxylic acid with a pair of carboxylic acid functional groups. The terminal carboxylic acid groups can react with primary amine groups in the presence of activators (e.g. EDC, or HATU) to form a stable amide bond.
Description
Succinic acid, or butanedioic acid, was first isolated as long ago as 1546 as a distillate from amber. It also occurs naturally in fungi and lichens, and it is a component of the citric acid cycle. Its chief reactions are oxidation to produce fumaric acid and condensation with ketones to produce unsaturated diacids. The commercialization of a biobased process for making succinic acid was announced in January.
Chemical properties
Succinic acid,C02H(CH2)2C02H, also known as butanedioic acid,butane diacid, and amber acid, is a colorless odorless prisms or white crystalline powder that melts at 185°C (364 of). Soluble in water and alcohol, it is used as a chemical intermediate, Succinic acid is used in lacquers,medicine,dyes,and as a taste modifier.
The Uses of Succinic acid
succinic acid is widely use as organic intermediates for the pharmaceutical, engineering plastics, resins etc.. For the synthesis of sedatives, contraceptives and cancer drugs in the pharmaceutical industry. In the chemical industry for the production of dyes, alkyd resin, glass fiber reinforced plastics, ion exchange resins and pesticides.
The Uses of Succinic acid
Succinic Acid is an acidulant that is commercially prepared by the hydrogenation of maleic or fumaric acid. it is a nonhygroscopic acid but is more soluble in 25°c water than fumaric and adipic acid. it has low acid strength and slow taste build-up; it is not a substitute for normal acidulants. it combines with proteins in modifying the plasticity of bread dough. it functions as an acidulant and flavor enhancer in relishes, beverages, and hot sausages.
The Uses of Succinic acid
Succinic Acid was identified in essential oil from Saxifraga stolonifera and has antibacterial activity.
Background
A water-soluble, colorless crystal with an acid taste that is used as a chemical intermediate, in medicine, the manufacture of lacquers, and to make perfume esters. It is also used in foods as a sequestrant, buffer, and a neutralizing agent. (Hawley's Condensed Chemical Dictionary, 12th ed, p1099; McGraw-Hill Dictionary of Scientific and Technical Terms, 4th ed, p1851)
Indications
For nutritional supplementation, also for treating dietary shortage or imbalance
Definition
ChEBI: An alpha,omega-dicarboxylic acid resulting from the formal oxidation of each of the terminal methyl groups of butane to the corresponding carboxy group. It is an intermediate metabolite in the citric acid cycle.
Definition
A crystalline carboxylic acid, HOOC(CH2)2COOH, that occurs in amber and certain plants. It forms during the fermentation of sugar (sucrose).
Production Methods
Succinic acid can be produced 1) by oxidation of Butanediol-1,4 with Nitric acid. 2) by oxidation of Tetrahydrofuran with Nitric acid. 3) by hydrogenation of Fumaric acid or Maleic acid.
Preparation
Succinic acid is derived from fermentation of ammonium tartrate. Hydrogenation of maleic acid, maleic anhydride, or fumaric acid produces good yields of succinic acid.
Biotechnological Production
Traditionally, succinic acid is produced by petrochemical synthesis using the precursor maleic acid. However, there are some microorganisms that are able to produce succinic acid (e.g. Actinobacillus succinogenes, Anaerobiospirillum succiniciproducens and Mannheimia succiniciproducens). Maximum product concentrations of 106 g.L-1 with a yield of 1.25 mol of succinic acid per mole of glucose and a productivity of 1.36 g.L-1.h-1 have been achieved by growing A. succinogenes on glucose . A high productivity of 10.40 g.L-1.h-1 has been reached with A. succinogenes growing on a complex medium with glucose in a continuous process with an integrated membrane bioreactor-electrodialysis process. In this process, the product concentration has been 83 g.L-1 . Moreover, metabolic engineering methods were used to develop strains (e.g. C. glutamicum, E. coli, S. cervisiae and Y. lipolytica) with high productivity and titer as well as low byproduct formation. For example, growing C. glutamicum strain DldhA-pCRA717 on a defined medium with glucose, a high productivity of 11.80 g.L-1.h-1 with a yield of 1.37 mol of succinic acid per mole of glucose and a titer of 83 g.L-1 has been reported after 7 h. An extended cultivation resulted in a product concentration of 146 g.L-1 after 46 h.
Synthesis Reference(s)
Canadian Journal of Chemistry, 56, p. 2269, 1978 DOI: 10.1139/v78-373
Synthesis, p. 709, 1984 DOI: 10.1055/s-1984-30945
General Description
White crystals or shiny white odorless crystalline powder. pH of 0.1 molar solution: 2.7. Very acid taste.
Air & Water Reactions
Slightly water soluble.
Reactivity Profile
Succinic acid reacts exothermically to neutralize bases, both organic and inorganic. Can react with active metals to form gaseous hydrogen and a metal salt. Such reactions are slow in the dry, but systems may absorb water from the air to allow corrosion of iron, steel, and aluminum parts and containers. Reacts slowly with cyanide salts to generate gaseous hydrogen cyanide. Reacts with solutions of cyanides to cause the release of gaseous hydrogen cyanide. May generate flammable and/or toxic gases and heat with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. May react with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), to generate flammable and/or toxic gases and heat. Can be oxidized exothermically by strong oxidizing agents and reduced by strong reducing agents. May initiate polymerization reactions.
Fire Hazard
Flash point data for Succinic acid are not available. Succinic acid is probably combustible.
Flammability and Explosibility
Non flammable
Biochem/physiol Actions
Succinic acid is a byproduct of anaerobic fermentation in microbes. It is a dicarboxylic acid and an intermediate in Kreb′s cycle. Polymorphism in succinate dehydrogenase leads to succinate accumulation. High levels of succinate impairs 2-oxoglutarate epigenetic signalling. Succinate levels may modulate tumor progression. Succinate inhibits histone demethylation and may contribute to epigenetic changes. Succinate is crucial for interleukin-1 β (IL-1β) synthesis during inflammation and immune signalling.
Biotechnological Applications
Succinic acid and its derivatives are used as flavoring agents for food and beverages. This acid could be used as feedstock for dyes, insecticides, perfumes, lacquers, as well as in the manufacture of clothing, paint, links, and fibers (McKinlay et al. 2007). Succinic acid is widely used in medicine as an antistress, antihypoxic, and immunity-improving agent, in animal diets, and as a stimulator of plant growth. It is also a component of bio-based polymers such as nylons or polyesters (Kamzolova et al. 2012b). Succinate esters are precursors for the known petrochemical products such as 1,4-butanediol, tetrahydrofuran, c-butyrolactone, and various pyrrolidinone derivatives (Bechthold et al. 2008).
Succinic acid production by Y. lipolytica was reported for the first time when it was grown on ethanol under aerobic conditions and nitrogen limitation. Succinic acid amount was 63.4 g/L as the major product of batch fermentation in this process. However, the disadvantage was low yield of succinic acid on ethanol (58 %), and a high cost of production (Kamzolova et al. 2009).
Kamzolova et al. developed a novel process for the production of succinic acid. It includes the synthesis of a-ketoglutaric acid by a thiamine-auxotrophic strain Y. lipolytica VKMY-2412 from n-alkanes, and subsequent oxidation of the acid by hydrogen peroxide to succinic acid. The concentration of succinic acid and its yield were found to be 38.8 g/L and 82.45 % of n-alkane consumed, respectively (Kamzolova et al. 2012b).
Succinic acid production was also studied by genetically modified strains using glucose and glycerol as substrates. Yuzbashev et al. constructed temperaturesensitive mutant strains with mutations in the succinate dehydrogenase encoding gene SDH1 by in vitro mutagenesis-based approach. Then, the mutants were used to optimize the composition of the media for selection of transformants with the deletion in the SDH2 gene. The defects of each succinate dehydrogenase subunit prevented the growth on glucose, but the mutant strains grew on glycerol and produced succinate in the presence of the buffering agent CaCO3. Subsequent selection of the strain with deleted SDH2 gene for increased viability was allowed to obtain a strain that is capable to accumulate succinate at the level of more than 450 g/L with buffering and more than 17 g/L without buffering. Therefore, a reduced succinate dehydrogenase activity can lead to an increased succinate production (Yuzbashev et al. 2010). Y. lipolytica is able to produce succinic acid at low pH values. High amounts of succinate can be achieved by genetic engineering (Otto et al. 2013).
Safety Profile
Moderately toxic by subcutaneous route. A severe eye irritant. Mutation data reported. When heated to decomposition it emits acrid smoke and irritating fumes.
Definition
Succinic acid is an alpha,omega-dicarboxylic acid resulting from the formal oxidation of each of the terminal methyl groups of butane to the corresponding carboxy group. It is an intermediate metabolite in the citric acid cycle. It has a role as a nutraceutical, a radiation protective agent, an anti-ulcer drug, a micronutrient and a fundamental metabolite. It is an alpha,omega-dicarboxylic acid and a C4-dicarboxylic acid. It is a conjugate acid of a succinate(1-).
Carcinogenicity
Monosodium succinate was given to groups of 50 male and 50 female Fischer 344 rats in drinking water at levels of 0%, 1%, or 2% for 2 years. No toxic lesion specifically caused by long-term administration of monosodium succinate was detected, and no dose-related increase was found in the incidence of tumors in any organ or tissue. The incidence of C-cell tumors of the thyroid gland of females that received 2% solution was apparently, but not significantly, higher than that in controls. Because C-cell tumors are commonly occurring spontaneous tumors in aging female rats of this strain and the incidence of C-cell tumors in the female control group was lower than that of historical controls for the testing laboratory, the authors concluded that this lesion was not treatment related.
Metabolism
Not Available
Purification Methods
Wash it with diethyl ether. Crystallise it from acetone, distilled water, or tert-butanol. Dry it under vacuum over P2O5 or conc H2SO4. Also purify it by conversion to the disodium salt which, after crystallisation from boiling water (charcoal), is treated with mineral acid to regenerate the succinic acid. The acid is then recrystallised and dried in a vacuum. [Beilstein 2 H 606, 2 IV 1908.]
Properties of Succinic acid
| Melting point: | 185 °C |
| Boiling point: | 235 °C |
| Density | 1.19 g/mL at 25 °C(lit.) |
| vapor pressure | 0-0Pa at 25℃ |
| FEMA | 4719 | SUCCINIC ACID |
| refractive index | n |
| Flash point: | >230 °F |
| storage temp. | 2-8°C |
| solubility | Soluble in ethanol, ethyl ether, acetone and methanol. Insoluble in toluene, benzene, carbon disulfide, carbon tetrachloride and petroleum ether. |
| form | Powder/Solid |
| pka | 4.16(at 25℃) |
| color | White to off-white |
| PH | 3.65(1 mM solution);3.12(10 mM solution);2.61(100 mM solution); |
| Odor | at 100.00 %. wormwood |
| Water Solubility | 80 g/L (20 ºC) |
| Merck | 14,8869 |
| BRN | 1754069 |
| Dielectric constant | 2.4(26℃) |
| Stability: | Stable. Substances to be avoided include strong bases, strong oxidizing agents. Combustible. |
| CAS DataBase Reference | 110-15-6(CAS DataBase Reference) |
| NIST Chemistry Reference | Butanedioic acid(110-15-6) |
| EPA Substance Registry System | Succinic acid (110-15-6) |
Safety information for Succinic acid
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05 |
| GHS Hazard Statements |
H318:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Succinic acid
| InChIKey | KDYFGRWQOYBRFD-UHFFFAOYSA-N |
Succinic acid manufacturer
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
You may like
-
 Succinic Acid 110 - 15 - 6 99%View Details
Succinic Acid 110 - 15 - 6 99%View Details
110 - 15 - 6 -
 Succinic acid 98%View Details
Succinic acid 98%View Details -
 Succinic acid 99%View Details
Succinic acid 99%View Details -
 Succinic acid CAS 110-15-6View Details
Succinic acid CAS 110-15-6View Details
110-15-6 -
 Succinic Acid CASView Details
Succinic Acid CASView Details -
 Succinic Acid CASView Details
Succinic Acid CASView Details -
 Succinic Acid chemicalView Details
Succinic Acid chemicalView Details
110-15-6 -
 Succinic Acid, 25kgView Details
Succinic Acid, 25kgView Details
110-15-6