Sebacic acid
Synonym(s):Decanedioic acid;Sebacic acid
- CAS NO.:111-20-6
- Empirical Formula: C10H18O4
- Molecular Weight: 202.25
- MDL number: MFCD00004440
- EINECS: 203-845-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 13:37:16
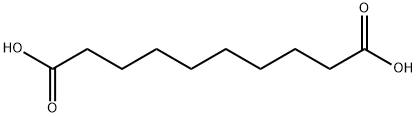
What is Sebacic acid?
Description
Sebacic acid is a dicarboxylic acid with structure (HOOC)(CH2)8(COOH), and is naturally occurring.
In its pure state it is a white flake or powdered crystal. The product is described as non-hazardous, though in its powdered form it can be prone to flash ignition (a typical risk in handling fine organic powders).
Sebaceus is Latin for tallow candle, sebum (tallow) is Latin for tallow, and refers to its use in the manufacture of candles. Sebacic acid is a derivative of castor oil, with the vast majority of world production occurring in China which annually exports over 20,000 metric tonnes, representing over 90 % of global trade of the product.
In the industrial setting, sebacic acid and its homologues such as azelaic acid can be used in plasticizers, lubricants, hydraulic fluids, cosmetics, candles, etc. Sebacic acid is also used as an intermediate for aromatics, antiseptics, and painting materials.
Chemical properties
White flaky crystals. Slightly soluble in water, soluble in alcohol and ether.
The Uses of Sebacic acid
Decanedioic acid was named by Thenard LJ (1802) from the Latin sebaceus(tallow candle) or sebum (tallow) in reference to its use in the manufacture of candles. Thenard LJ isolated this compound from distillation products of beef tallow. In 1954, it was reported that it was produced in excess of 10,000 tons annually by alkali fission of castor oil. Sebacic acid and its derivatives, as azelaic acid, have a variety of industrial uses as plasticizers, lubricants, diffusion pump oils, cosmetics, candles, etc. It is also used in the synthesis of polyamide, as nylon, and of alkyd resins. An isomer, isosebacic acid, has several applications in the manufacture of vinyl resin plasticizers, extrusion plastics, adhesives, ester lubricants, polyesters, polyurethane resins and synthetic rubber.
The Uses of Sebacic acid
Sebacic Acid is a urinary metabolite that has been identified as an anti-fatigue biomarker.
Preparation
Sebacic acid is normally made from castor oil, which is essentially glycerol
triricinoleate. The castor oil is heated with sodium hydroxide at about 250??e.
This treatment results in saponification of the castor oil to ricinoleic acid
which is then cleaved to give 2-octanol and sebacic acid:
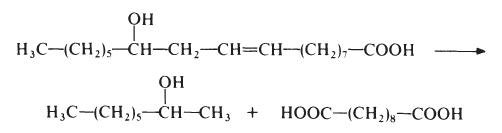
This process results in low yields of sebacic acid (about 50% based on the castor oil) but, nevertheless, other routes have not proved competitive. Sebacic acid is a colourless crystalline solid, m.p. 134??.
Definition
ChEBI: Sebacic acid is an alpha,omega-dicarboxylic acid that is the 1,8-dicarboxy derivative of octane. It has a role as a human metabolite and a plant metabolite. It is an alpha,omega-dicarboxylic acid and a dicarboxylic fatty acid. It is a conjugate acid of a sebacate(2-) and a sebacate. It derives from a hydride of a decane.
General Description
White granular powder. Melting point 153°F. Slightly soluble in water. Sublimes slowly at 750 mm Hg when heated to melting point.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
Sebacic acid reacts exothermically to neutralize bases, both organic and inorganic. May react rapidly with aqueous solutions containing a chemical base and dissolve as the neutralization generates a soluble salt. Can react with active metals to form gaseous hydrogen and a metal salt. Such reactions are slow in the dry, but systems may absorb enough water from the air to allow corrosion of iron, steel, and aluminum parts and containers. Reacts slowly with cyanide salts to generate gaseous hydrogen cyanide. Reacts with solutions of cyanides to cause the release of gaseous hydrogen cyanide. May generate flammable and/or toxic gases and heat with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. May react with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), to generate flammable and/or toxic gases and heat. Can be oxidized exothermically by strong oxidizing agents and reduced by strong reducing agents. May initiate polymerization reactions.
Fire Hazard
Flash point data for Sebacic acid are not available. Sebacic acid is probably combustible.
Flammability and Explosibility
Not classified
Purification Methods
Purify sebacic acid via the disodium salt which, after crystallisation from boiling water (charcoal), is again converted to the free acid. The free acid is crystallised repeatedly from hot distilled water or from Me2CO/pet ether and dried under vacuum. [Beilstein 2 IV 2078.]
Properties of Sebacic acid
| Melting point: | 133-137 °C (lit.) |
| Boiling point: | 294.5 °C/100 mmHg (lit.) |
| Density | 1.21 |
| vapor pressure | 1 mm Hg ( 183 °C) |
| refractive index | 1.422 |
| Flash point: | 220 °C |
| storage temp. | Store below +30°C. |
| solubility | ethanol: 100 mg/mL |
| form | Powder or Granules |
| pka | 4.59, 5.59(at 25℃) |
| color | White to off-white |
| Odor | monoclinic prismatic tablets, wh. powd., fatty acid odor |
| Water Solubility | 1 g/L (20 ºC) |
| Merck | 14,8415 |
| BRN | 1210591 |
| Stability: | Stable. Combustible. Incompatible with strong oxidizing agents, bases, reducing agents. |
| CAS DataBase Reference | 111-20-6(CAS DataBase Reference) |
| NIST Chemistry Reference | Decanedioic acid(111-20-6) |
| EPA Substance Registry System | Sebacic acid (111-20-6) |
Safety information for Sebacic acid
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P280:Wear protective gloves/protective clothing/eye protection/face protection. P304+P340:IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P405:Store locked up. |
Computed Descriptors for Sebacic acid
| InChIKey | CXMXRPHRNRROMY-UHFFFAOYSA-N |
Sebacic acid manufacturer
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 SEBACIC ACID 99%View Details
SEBACIC ACID 99%View Details -
 Sebacic Acid pure CAS 111-20-6View Details
Sebacic Acid pure CAS 111-20-6View Details
111-20-6 -
 Sebacic acid CAS 111-20-6View Details
Sebacic acid CAS 111-20-6View Details
111-20-6 -
 Sebacic acid CAS 111-20-6View Details
Sebacic acid CAS 111-20-6View Details
111-20-6 -
 Sebacic Acid CAS 111-20-6View Details
Sebacic Acid CAS 111-20-6View Details
111-20-6 -
 Sebacic Acid CASView Details
Sebacic Acid CASView Details -
 Sebacic Acid CASView Details
Sebacic Acid CASView Details -
 Sebacic acid 95.00% CAS 111-20-6View Details
Sebacic acid 95.00% CAS 111-20-6View Details
111-20-6