RHENIUM
Synonym(s):;Rhenium;Rhenium element;Rhenium nitrate solution
- CAS NO.:7440-15-5
- Empirical Formula: Re
- Molecular Weight: 186.21
- MDL number: MFCD00011195
- EINECS: 231-124-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:08:52
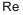
What is RHENIUM?
Chemical properties
gray powder
Chemical properties
Depending on the process used to isolate and process it,
rhenium may appear as a brown-black powder or a silvery
white solid metal. Rhenium is among the least common of the
natural elements comprising 0.5–1 ppb of earth’s crust; it
generally occurs as a trace element in molybdenite, columbite,
gadolinite, and platinum ores. A sulfide mineral of
rhenium, rhenite exists but is very rare.
There are two naturally occurring isotopes, 185 (37%),
which is stable, and 187 (63%), which has a halftime of 1011
years, and several synthetic radioisotopes whose half-lives
range from ,<1 μs to 2×105 year. Rhenium has 11
valence states that range from 0 to 7.
Physical properties
Rhenium ranges in color from silvery-white to gray to a black powder. It is a rather denseelement. As a refined metal, rhenium is ductile, but because it is rather rare, its properties havenot found many uses. Rhenium does have the widest range of valences. In addition to its commonvalences of 4, 6, and 7, it also has the uncommon valences of 2, –1, and –7.
Rhenium has a high melting point of 3,180°C, a boiling point of 5,627°C, and a densityof 21.04 g/cm3.
Isotopes
There are 45 isotopes of rhenium. Only one of these is stable: Re-185, whichcontributes 37.40% to the total amount of rhenium found on Earth. Re-187, which isradioactive with a very long half-life of 4.35×10+10 years, contributes 62.60% to rhenium’sexistence on Earth. The remaining 43 isotopes are radioactive with relatively shorthalf-lives and are artificially manufactured.
Origin of Name
Derived from the Latin word Rhenus, which stands for the Rhine River in Western Europe.
Occurrence
Rhenium is the 78th most common element found on Earth, which makes it somewhatrare. During the early twentieth century, it required the processing of about a 1,000 poundsof earth to secure just one pound of rhenium, resulting in a price of about $10,000 per gram.Thus, there were few uses for rhenium. Later in the century, improved mining and refiningtechniques reduced the price. Today, the United States produces about 1,000 pounds of rheniumper year, and the world’s total estimated supply is only about 400 tons.
The main sources of rhenium are the molybdenite and columbite ores. Some rhenium isrecovered as a by-product of the smelting of copper sulfide (CuS) ores. Molybdenum sulfide(MoS2) is the main ore and is usually associated with igneous rocks and, at times, metallic-likedeposits. Molybdenite is found in Chile, as well as in the states of New Mexico, Utah, andColorado in the United States.
History
Discovery of rhenium is generally attributed to Noddack, Tacke, and Berg, who announced in 1925 they had detected the element in platinum ores and columbite. They also found the element in gadolinite and molybdenite. By working up 660 kg of molybdenite they were able in 1928 to extract 1 g of rhenium. The price in 1928 was $10,000/g. Rhenium does not occur free in nature or as a compound in a distinct mineral species. It is, however, widely spread throughout the Earth’s crust to the extent of about 0.001 ppm. Commercial rhenium in the U.S. today is obtained from molybdenite roaster-flue dusts obtained from copper-sulfide ores mined in the vicinity of Miami, Arizona, and elsewhere in Arizona and Utah. Some molybdenites contain from 0.002 to 0.2% rhenium. It is estimated that in 1999 about 16,000 kg of rhenium was being produced. The total estimated world reserves of rhenium is 11,000,000 kg. Natural rhenium is a mixture of two isotopes, one of which has a very long half-life. Thirty-nine other unstable isotopes are recognized. Rhenium metal is prepared by reducing ammonium perrhenate with hydrogen at elevated temperatures. The element is silvery white with a metallic luster; its density is exceeded by that of only platinum, iridium, and osmium, and its melting point is exceeded by that of only tungsten and carbon. It has other useful properties. The usual commercial form of the element is a powder, but it can be consolidated by pressing and resistance-sintering in a vacuum or hydrogen atmosphere. This produces a compact shape in excess of 90% of the density of the metal. Annealed rhenium is very ductile, and can be bent, coiled, or rolled. Rhenium is used as an additive to tungsten and molybdenum-based alloys to impart useful properties. It is widely used for filaments for mass spectrographs and ion gages. Rhenium-molybdenum alloys are superconductive at 10 K. Rhenium is also used as an electrical contact material as it has good wear resistance and withstands arc corrosion. Thermocouples made of Re-W are used for measuring temperatures up to 2200°C, and rhenium wire has been used in photoflash lamps for photography. Rhenium catalysts are exceptionally resistant to poisoning from nitrogen, sulfur, and phosphorus, and are used for hydrogenation of fine chemicals, hydrocracking, reforming, and disproportionation of olefins. Rhenium has recently become especially important as a catalyst for petroleum refining and in making super-alloys for jet engines. Rhenium costs about $16/g (99.99% pure). Little is known of its toxicity; therefore, it should be handled with care until more data are available.
Characteristics
Rhenium is one of the transition elements, which range from metals to metal-like elements.Its chemical and physical properties are similar to those of technetium, which is aboveit in the periodic table. It is not very reactive. When small amounts are added to molybdenum,it forms a unique type of semiconducting metal. It is also noncorrosive in seawater.
The Uses of RHENIUM
Small quantities of rhenium are alloyed with iron to form steel that is both hard and resistantto wear and high-temperatures. Because of its high melting point, rhenium is used inmany applications where long-wearing, high-temperature electrical components are required,such as electrical contacts and switches and high-temperature thermocouples. This physicalquality makes rhenium alloys ideal for use in rocket and missile engines. It is also used to formthe filaments in photographic flash lamps.
Rhenium’s isotope (187Re) has a very long half-life and decays by both beta and alpharadiation at a very steady rate. This factor makes it useful as a standard to measure the age ofthe universe.
The Uses of RHENIUM
Electron tube and semiconductor applications, in alloys for electrical contacts, as catalyst; possibly in high tempereture thermocouples and to improve the workability of tungsten and molybdenum alloys; plating jewelry, medical instruments, high vac equipment, mirror backings.
The Uses of RHENIUM
Rhenium, 1% on 2.5mm alumina spheres is a heterogeneous catalyst used in synthetic chemistry. It is also tested for ammonia synthesis.
Definition
Metallic element, atomic number 75, group VIIB of the periodic table, aw 186.207; valences = ?1, 1 through 7; 4, 6, and 7 are most common, the last being the most stable. There are two isotopes.
Definition
A rare silvery transition metal that usually occurs naturally with molybdenum (it is extracted from flue dust in molybdenum smelters). The metal is chemically similar to manganese and is used in alloys and catalysts. Symbol: Re; m.p. 3180°C; b.p. 5630°C; r.d. 21.02 (20°C); p.n. 75; r.a.m. 186.207.
Definition
rhenium: Symbol Re. A silvery white metallic transition element;a.n. 75; r.a.m. 186.2; r.d. 20.53; m.p.3180°C; b.p. 5627 (estimated)°C. Theelement is obtained as a by-productin refining molybdenum, and is usedin certain alloys (e.g. rhenium–molybdenum alloys are superconducting).The element forms a numberof complexes with oxidationstates in the range 1–7. It was discoveredby Walter Noddack (1893–1960)and Ida Tacke (1896–1978) in 1925.
Production Methods
Among the compounds that can be formed with rhenium are sulfides, fluorides, chlorides, bromides, iodides, and oxides. Rhenium(VII) oxide, Re2O7, is the most stable oxide of rhenium. It is formed from rhenium metal powder or other rhenium oxides in dry air or an oxygen atmosphere above 350° °C. Re2O7 is readily soluble in water, forming perrhenic acid, HReO4, which forms salts (MReO4) such as ammonium perrhenate (NH4ReO4). This is an important starting material, which can be reduced to Re metal and used for the production of many other rhenium compounds. Rhenium can also form organometallic compounds such as carbonyls (e.g., Re2(CO)10 and organorhenium compounds such as hexamethylrhenium, Re(CH3)6.
Hazard
Flammable in powder form.
Hazard
Rhenium is flammable in powder form. Rhenium dust and powder and many of its compoundsare toxic when inhaled or ingested.
Flammability and Explosibility
Non flammable
Industrial uses
The outstanding properties of rhenium and rhenium alloys suggest their use in many specialized applications.Rhenium vs. tungsten thermocouples can be used for temperature measurement and control to approximately 2200 C, whereas previous thermocouple use was limited to temperatures below 1750 C. Indeed, 74% W 26% Re vs. W thermocouples can be used to temperatures of at least 2750 C with a high emf output ensuring accurate precise temperature measurements with excellent reproducibility and reliability.
Rhenium is now widely used for filaments for mass spectrographs and for ion gauges for measuring high vacuum. Its ductility, chemical properties, including the fact that it does not react with carbon to form a carbide, and its emission characteristics make it superior to tungsten for these applications.
Rhenium has received considerable acclaim as an electrical contact material. It possesses excellent resistance to wear as well as arc erosion. Furthermore, the contact resistance of rhenium is extremely stable because of its good corrosion resistance in addition to the fact that possible formation of an oxide film on the contacts would not cause any appreciable change in the contact resistance since the resistivity of the oxide is almost the same as that of the metal. Extensive tests for some types of make-andbreak switching contacts has shown rhenium to have 20 times the life of platinum palladium contacts currently in use.
Properties of RHENIUM
| Melting point: | 3180 °C (lit.) |
| Boiling point: | 5596 °C (lit.)
5627 °C (lit.) |
| Density | 21.02 g/cm3 (lit.) |
| storage temp. | Flammables area |
| solubility | insoluble in HCl |
| form | wire |
| color | Silver-gray |
| Specific Gravity | 21.04 |
| Water Solubility | Insoluble in water. |
| Merck | 13,8261 |
| Exposure limits | ACGIH: TWA 1 mg/m3 OSHA: TWA 15 mg/m3; TWA 5 mg/m3 |
| CAS DataBase Reference | 7440-15-5(CAS DataBase Reference) |
| EPA Substance Registry System | Rhenium (7440-15-5) |
Safety information for RHENIUM
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05 |
| GHS Hazard Statements |
H290:Corrosive to Metals H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P234:Keep only in original container. P280:Wear protective gloves/protective clothing/eye protection/face protection. P363:Wash contaminated clothing before reuse. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for RHENIUM
| InChIKey | WUAPFZMCVAUBPE-UHFFFAOYSA-N |
RHENIUM manufacturer
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 Rhenium CAS 7440-15-5View Details
Rhenium CAS 7440-15-5View Details
7440-15-5 -
 Rhenium CAS 7440-15-5View Details
Rhenium CAS 7440-15-5View Details
7440-15-5 -
 Rhenium wire, 0.1mm (0.004 in.) dia., Annealed CAS 7440-15-5View Details
Rhenium wire, 0.1mm (0.004 in.) dia., Annealed CAS 7440-15-5View Details
7440-15-5 -
 Rhenium wire, 0.25mm (0.01 in.) dia. CAS 7440-15-5View Details
Rhenium wire, 0.25mm (0.01 in.) dia. CAS 7440-15-5View Details
7440-15-5 -
 Rhenium wire, 0.25mm (0.01 in.) dia. CAS 7440-15-5View Details
Rhenium wire, 0.25mm (0.01 in.) dia. CAS 7440-15-5View Details
7440-15-5 -
 Rhenium CAS 7440-15-5View Details
Rhenium CAS 7440-15-5View Details
7440-15-5 -
 Rhenium CAS 7440-15-5View Details
Rhenium CAS 7440-15-5View Details
7440-15-5 -
 Rhenium standard for AAS CASView Details
Rhenium standard for AAS CASView Details