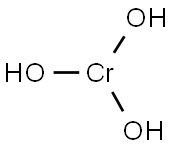
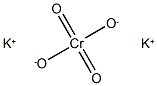
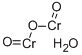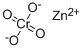potassium hydroxyoctaoxodizincatedichromate(1-)
- CAS NO.:11103-86-9
- Empirical Formula: CrH2KO5Zn-3
- Molecular Weight: 238.49
- EINECS: 234-329-8
- Update Date: 2024-12-18 14:08:52
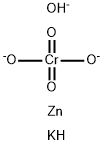
What is potassium hydroxyoctaoxodizincatedichromate(1-) ?
Description
Potassium hydroxyoctaoxodizincatedichromate (zinc potassium
chromate) is a green-yellow, odorless solid. It is one of
a number of chromate compounds used as inhibitors of rust
and metal corrosion. While the counterions zinc and potassium
lend unique physical and chemical characteristics to the
compound, characteristics such as acidity and solubility, it is
the chromate ion that gives rise to most critical properties, both
industrially and toxicologically.
Ionic chromium (Cr) is typically found in either the hexavalent
(Cr(VI)) or trivalent (Cr(III)) oxidation states. Chromate
ion (CrO4--2) is among the most common Cr(VI) compounds.
Other common Cr(VI) compounds include dichromate
(Cr2O7-2), and chromium trioxide (CrO3).
In aqueous solution, Cr(VI) exists as hydrochromate
(HCrO4-), CrO4-2, or Cr2O7-2. Dichromate is the dimer of
chromate. The three species exist in equilibrium with one
another, the proportion of each being concentration and pH
dependent. Chromate ion is the predominant form in dilute
solutions at neutral pH. Under these conditions, very little
dichromate exists and the ratio of chromate to hydrochromate
is about 3 to 1. Hydrochromate becomes the dominant
species of Cr(VI) when the pH becomes mildly acidic (<6).
In concentrated solutions of Cr(VI) in strongly acidic conditions,
dichromate becomes the dominant species.
Specific data and literature regarding the toxicity and characteristics
of zinc potassium chromate are limited. On the other
hand, literature relating to the health and environmental effects
of chromates and other Cr(VI) compounds is much more
extensive. Most experimentalists and regulatory agencies make
the assumption that chromates, dichromates, and indeed,
all Cr(VI) compounds behave similarly in biological systems.For this reason, Cr(IV) compounds and their biological
behavior are often discussed as a class; in the same way, Cr(III)
compounds are often lumped together.
When data specific to zinc potassium chromate are available,
it is presented. In other cases, generalizations available
from studies of chromates as a class or Cr(VI) compounds will
be presented.
The Uses of potassium hydroxyoctaoxodizincatedichromate(1-)
Zinc potassium chromate is one of several chromates used as anticorrosive agents in the formulation of coatings and primers. Chromates are widely used as inhibitors of corrosion and rust because of their unique ability to react at the metal coating interface to inhibit corrosion, especially galvanic couple corrosion (a chemical reaction that involves electron exchange between different metals). For example, using stainless steel screws on an aluminum part provides a high potential for electron transfer (galvanic couple corrosion). Even when present in very low concentrations, chromate has the unique ability to actively suppress electron transfer at both cathodic and anodic sites when different metallic parts are in contact. Active protection against rust depends on the ability of the inhibitor to migrate to the exposed surface once the protective coating has been scratched or damaged. Inhibitors dissolve in water and migrate to the exposed surface. If the inhibitor’s solubility is too great it may be washed away, if the solubility is too low the inhibitor will have low activity. According to the literature, strontium chromate has an ideal solubility (1.06 g l-1); zinc potassium chromate has similar solubility (0.5–1.5 g l-1) and is a very effective inhibitor. No single nonchrome inhibitor tested thus far acts in this way.
The Uses of potassium hydroxyoctaoxodizincatedichromate(1-)
Potassium Hydroxyoctaoxodizincatedichromate is a corrosion-inhibiting zinc pigment used to protect steel and other metals.
Safety Profile
Confirmed carcinogen. Mutation data reported. When heated to decomposition it emits toxic fumes of ZnO and K2O. Used as a corrosion inhibiting pigment and in steel priming. See also CHROMIUM COMPOUNDS and ZINC COMPOUNDS.
Environmental Fate
Chromium (both trivalent and hexavalent) enters the environment
from numerous natural and anthropogenic sources.
The health hazards of environmental exposure depend on the
oxidation state, with Cr(VI) being most toxic. Cr(VI) contamination
of groundwater typically occurs from industrial sources
such as electroplating or corrosion protection. Contamination
of surface water is commonly the result of particulate
discharges into the air from manufacturing and cooling towers,
with the particulates ultimately settling to either soil or surface
waters. For years, Cr(VI) was thought to arise environmentally
only as an industrial pollutant but recently unpolluted ground
and surface waters have been found to contain Cr(VI) in
concentrations that exceed the World Health Organization
limit for drinking water (50 mg l-1).
Much of the Cr(VI) in the environment is ultimately reduced
to the less toxic Cr(III). The reduction may be mediated by
various reducing agents such as sulfide compounds, and divalent
iron (Fe(II)). In addition, organic matter (e.g., humic acid and fulvic acid) in water or soil may mediate the reduction
process. Microbial processes also convert Cr(VI) to Cr(III).
Toxicity evaluation
Chromate is taken up by cells through sulfate channels. Once
in the cell it can cause both oxidative and nonoxidative forms of DNA damage. The most dominant form of damage is Cr-
DNA adduct formation, a process that occurs in vitro at Cr(VI)
concentrations of less than 2 mM. The process involves reduction
of Cr(VI) to Cr(III) during the formation of either binary
(Cr(III)–DNA) or ternary (ligand–Cr(III)–DNA) adducts. In the
ternary adducts, the ligand can be ascorbate (Asc), glutathione
(GSH), cysteine, or histidine. In in vitro studies, binary adducts
were found to account for 75–95% of the total DNA-bound Cr.
Asc, GSH, and cysteine represent the three most important
biological reducers of Cr(VI) and are key to formation of the
ternary adducts. These ternary adducts are strongly mutagenic.
When reducing agents such as Asc are depleted, Cr(VI)
reduction leads to the formation of reactive oxygen species
which may lead to oxidative damage of DNA.
It has also been proposed that depletion of Asc (as via
oxidation by Cr(VI)) may impede removal of repressive DNA
(methylated cytosine-phosphate-guanine) and histone H3
marks which may modulate gene expression. Chromium(VI)
also causes the formation of protein–Cr(III)–DNA cross-links.
The formation of these adducts is rare but it has been suggested
that they may influence gene specific expression.
The damage induced by Cr(VI) leads to dysfunctional DNA
replication and transcription, aberrant cell cycle checkpoints,
poorly regulated DNA repair mechanisms, inflammatory
responses, and the disruption of regulatory genes responsible
for the normal balance between cellular survival and death.
Disruption of these processes result in neoplastic progression.
Properties of potassium hydroxyoctaoxodizincatedichromate(1-)
| Water Solubility | 500mg/L at 20℃ |
| EPA Substance Registry System | Potassium zinc chromate hydroxide (11103-86-9) |
Safety information for potassium hydroxyoctaoxodizincatedichromate(1-)
Computed Descriptors for potassium hydroxyoctaoxodizincatedichromate(1-)
potassium hydroxyoctaoxodizincatedichromate(1-) manufacturer
Trinity Pigment Industries
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate N-octanoyl benzotriazole 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
You may like
-
 11103-86-9 Potassium hydroxyoctaoxodizincatedichromate(1-) 99%View Details
11103-86-9 Potassium hydroxyoctaoxodizincatedichromate(1-) 99%View Details
11103-86-9 -
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1