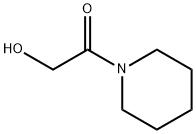
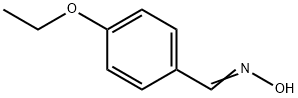
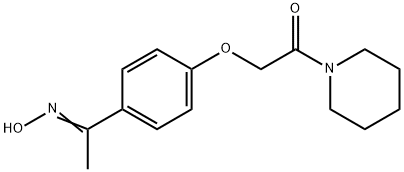
Pifoxime
- CAS NO.:31224-92-7
- Empirical Formula: C15H20N2O3
- Molecular Weight: 276.33
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-19 20:33:22
What is Pifoxime?
Originator
Flamanil ,Salvoxyl-Wander, France ,1975
Manufacturing Process
(A) Preparation of p-Acetylphenoxyacetic Acid: p-Hydroxy-acetophenone is treated with chloroacetic acid in aqueous solution in the presence of sodium hydroxide. The desired acid is then isolated from its sodium salt in a total yield of 80 to 82%, excess of p-hydroxy-acetophenone having been extracted with methylene chloride.
(B) Preparation of Methyl p-Acetylphenoxy-Acetate: A mixture of 80 g of the acid obtained in (A) and 200 ml of methyl alcohol in 600 ml of dichloromethane is refluxed in the presence of sulfuric acid. The desired ester is isolated in accordance with a method known per se, and recrystallized. When the refluxing period is 12 hours, the ester is obtained with a yield of 70%. When the refluxing period is 18 hours, the yield for this ester is 85%.
(C) Preparation of N-(p-Acetylphenoxy-Acetyl)-Piperidine: The ester from (B) is refluxed for 8 hours with 2.5 mols of thoroughly dried piperidine. Then 1 volume of water is added and the product is left to crystallize in the cold. The desired amide is obtained in an 80% yield.
(D) Preparation of N-(p-[1-Isonitrosoethyl]-Phenoxy-Acetyl)-Piperidine: The amide from (C) is refluxed for 5 hours with technical (98%) hydroxylamine and alcohol denatured with methanol. The desired product is obtained in a 75% yield.
In semiindustrial synthesis, to achieve better yields, it is possible to omit (A), by directly preparing the ester (B) by reaction of p-hydroxy acetophenone on ethyl 2-bromoacetate in the presence of potassium carbonate in butanone. The yield of ester is 90%, and elimination of excess of phydroxyacetophenone is effected by washing with sodium hydroxide.
Therapeutic Function
Antiinflammatory
Properties of Pifoxime
| Boiling point: | 494.7±30.0 °C(Predicted) |
| Density | 1.18±0.1 g/cm3(Predicted) |
| pka | 11.50±0.70(Predicted) |
Safety information for Pifoxime
Computed Descriptors for Pifoxime
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 4-Hydrazinobenzoic acid 3,4-Dibenzyloxybenzaldehyde Electrolytic Iron Powder 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 4-HYDROXY BENZYL ALCOHOL 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) S-2-CHLORO PROPIONIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 3-(Hydroxymethyl)benzoate N-Boc-2-chloroethylamine 1-Bromo-2-methoxy-3-nitrobenzene N-Methyl-3-cyclopenten-1-amine 2-Bromo-3-hydroxybenzaldehyde 1H-indazole-5-carboxamideRelated products of tetrahydrofuran
You may like
-
 7441-43-2 98%View Details
7441-43-2 98%View Details
7441-43-2 -
 1260741-78-3 6-Bromo-3-iodo-1-methyl-1H-indazole 98%View Details
1260741-78-3 6-Bromo-3-iodo-1-methyl-1H-indazole 98%View Details
1260741-78-3 -
 (3-Benzyloxypropyl)triphenyl phosphonium 98%View Details
(3-Benzyloxypropyl)triphenyl phosphonium 98%View Details
54314-85-1 -
 4-bromo-3,5-dimethylbenzenesulfonyl chloride 1581266-79-6 98%View Details
4-bromo-3,5-dimethylbenzenesulfonyl chloride 1581266-79-6 98%View Details
1581266-79-6 -
 2490430-37-8 98%View Details
2490430-37-8 98%View Details
2490430-37-8 -
 N-(5-Amino-2-methylphenyl)acetamide 5434-30-0 98%View Details
N-(5-Amino-2-methylphenyl)acetamide 5434-30-0 98%View Details
5434-30-0 -
 124371-59-1 98%View Details
124371-59-1 98%View Details
124371-59-1 -
 53857-52-2 98%View Details
53857-52-2 98%View Details
53857-52-2