Phenmetrazine
- CAS NO.:134-49-6
- Empirical Formula: C11H15NO
- Molecular Weight: 177.24
- MDL number: MFCD00864487
- EINECS: 205-143-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:15:32
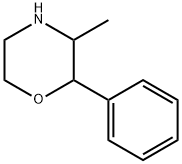
What is Phenmetrazine?
Absorption
Readily absorbed from the gastro-intestinal tract and buccal mucosa.
Toxicity
Adult monkeys have an LD50 of 15 to 20 mg/kg, whereas for young monkeys the LD50 is only 5 mg/kg. Symptoms of overdose include acute central nervous system stimulation, cardiotoxicity causing tachycardia, arrhythmias, hypertension, and cardiovascular collapse. Whilst some patients show signs of toxicity at blood concentrations of 20 μg/L, chronic abusers of amphetamine have been known to have blood concentration of up to 3000 μg/L.
Originator
Preludin, Boehringer Ingelheim, US ,1956
The Uses of Phenmetrazine
Anorexic.
Background
A sympathomimetic drug used primarily as an appetite depressant. Its actions and mechanisms are similar to dextroamphetamine.
Indications
Used as an anorectic in the treatment of obesity.
Definition
ChEBI: Phenmetrazine is a member of the class of morpholines that is morpholine substituted with a phenyl group at position 2 and a methyl group at position 3. It has a role as a metabolite and a sympathomimetic agent. It derives from a morpholine.
Manufacturing Process
10 grams of β-phenyl-α-methyl-β,β'-dihydroxy-diethylamine hydrochloride (produced by hydrogenation in the presence of palladium and charcoal of βphenyl-α-methyl-β-keto-β'-hydroxy-N-benzyl-diethylamine hydrochloride obtained from bromopropiophenone by reacting with benzyl-ethanolamine), are warmed with 10% hydrochloric acid for 6 hours on a water bath.After working up in the usual manner, the hydrochloride of the 2-phenyl-3methyl-morpholine crystallizes out from methanolic hydrochloric acid and acetone, MP = 182°C, according to US Patent 2,835,669.
brand name
Preludin (Boehringer Ingelheim);Anorex;Bromadryl;Emagrin;Filon;Gratsidin;Marsin;Neo-zine;Prelazine.
Therapeutic Function
Antiobesity
World Health Organization (WHO)
Phenmetrazine, a sympathomimetic amine, was introduced in 1956 for use as an anorexic agent. Although preparations remain available, the use of phenmetrazine is no longer indicated for the treatment of obesity. Moreover, since it has been subject to abuse, and because dependence can occur, it is now controlled under Schedule II of the 1971 Convention on Psychotropic Substances. (Reference: (UNCPS2) United Nations Convention on Psychotropic Substances (II), , , 1971)
Pharmacokinetics
Phenmetrazine is a sympathomimetic drug used primarily as an appetite depressant. Its actions and mechanisms are similar to dextroamphetamine. Amphetamines are non-catecholamine sympathomimetic amines with CNS stimulant activity. Phenmetrazine was originally sold under the tradename Preludin as an anorectic. It has since been removed from the market. It is by some considered to have a greater potential for addiction than the amphetamines, and has been abused in many countries, for example Sweden.
Pharmacology
Phenmetrazine is a sympathomimetic drug used primarily as an appetite depressant. Its actions and mechanisms are similar to dextroamphetamine. Amphetamines are non-catecholamine sympathomimetic amines with CNS stimulant activity. Phenmetrazine was originally sold under the tradename Preludin as an anorectic. It has since been removed from the market. It is by some considered to have a greater potential for addiction than the amphetamines, and has been abused in many countries, for example Sweden.
Metabolism
Primarily hepatic (via CYP3A and CYP2D6). Resistant to metabolism by monoamine oxidase. Metabolism involves deamination to para-hydroxyamphetamine and phenylacetone; this latter compound is subsequently oxidize to benzoic acid and excreted as glucuronide or glycine (hippuric acid) conjugate. Smaller amounts of amphetamine are converted to norephedrine by oxidation.
Properties of Phenmetrazine
| Boiling point: | bp12 138-140°; bp1 104° |
| Density | 1.0290 (rough estimate) |
| refractive index | 1.5302 (estimate) |
| pka | pKa 8.45 (Uncertain) |
| EPA Substance Registry System | Morpholine, 3-methyl-2-phenyl- (134-49-6) |
Safety information for Phenmetrazine
Computed Descriptors for Phenmetrazine
New Products
4-Fluorophenylacetic acid 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate (6-METHYL-[1,3]DITHIOLO[4,5-b]QUINOXALIN-2-ONE INDAZOLE-3-CARBOXYLIC ACID 4-IODO BENZOIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran


You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1