Perfluorooctyl iodide
Synonym(s):1-Iodoperfluorooctane;Heptadecafluorooctyl iodide;Perfluorooctyl iodide
- CAS NO.:507-63-1
- Empirical Formula: C8F17I
- Molecular Weight: 545.96
- MDL number: MFCD00001064
- EINECS: 208-079-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-04-24 16:33:08
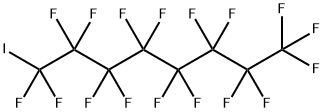
What is Perfluorooctyl iodide?
Chemical properties
clear light pink liquid after melting
The Uses of Perfluorooctyl iodide
Perfluorooctyl Iodide is a perfluoroalkyl iodide used in organocatalysis via substrate activation by halogen bonding. Perfluorooctyl Iodide is a potential candidate substitute for banned Halon fire extinguishers.
The Uses of Perfluorooctyl iodide
Heptadecafluoro-1-iodooctane has been used as monodentate donor to investigate anion receptor complexes of mono-, bi- and tridentate donors with a variety of anions in the gas phase using both experimental and theoretical approaches.
Reactivity Profile
1H,1H,2H,2H-1Iodide-Perfluorodecane was synthesized using perfluorooctyl iodide and ethylene as materials, then reacted with sodium methoxide /methanol to gain 1H,1H,2H-Heptadecafluorodecene. Influence factors such as the kinds of solvents, raw material configuration, concentration, and time used in the process of dehydrogen iodide were studied mainly. Optimization was using 1,1,2-Trichloro-1,2,2-trifluoroethane as solvent, sodium methoxide /methanol (2mol/L ), 1H,1H,2H,2H-1Iodide-Perfluorodecane( M1 ): sodium methoxide( M2 ) =1:2, reacted 12 hours, the yield of 1H,1H,2H-Heptadecafluorodecene was up to 80%[4-5].
Cytotoxicity
Exposure of H295R cells to Perfluorooctyl iodide (PFOI) (<100 μM) and Perfluorooctanoic acid (PFOA) (<100 μM) did not cause significant toxicity within the concentration ranges tested (p > 0.05). However, 125 μM PFOI and PFOA induced significant toxicity in H295R cells (p < 0.05). No obvious morphological alterations were observed in the cells treated with PFOI or PFOA at levels lower than 100 μM upon microscopic examination[1].
Biochem/physiol Actions
Levels of aldosterone production, cortisol, and 17β-estradiol were elevated significantly, and the level of testosterone generation decreased upon treatment with 100 μM PFOI. Similar to the effect induced by forskolin (AC activator), expression of all 10 genes involved in synthesizing steroid hormones was upregulated significantly upon exposure to 100 μM PFOI. PFOA had no effect on steroid hormone production or steroidogenic gene expression, even though it is highly structurally similar to PFOI. Therefore, the terminal -CF2I group in PFOI could be a critical factor for the mediation of steroidogenesis. PFOI increased AC activity and cAMP levels in H295R cells, which implied an underlying mechanism for the disturbance of steroidogenesis. These data suggest that PFOI may act as an AC activator, thereby stimulating steroidogenesis by activating a cAMP signaling pathway[3].
Safety Profile
Slightly toxic by intravenous route. When heated to decomposition it emits toxic vapors of Fí and Ií.
Environmental considerations
Production of perfluorooctyl iodide (PFOI) has great commercial value. PFOI is a potential estrogenic compound. Studies have shown that the concentration of PFOI is relatively higher in the air and soil than the concentrations of the other derivatives of Perfluorinated iodine alkanes (PFIs). In addition, the degradation of PFIs can cause the formation of other Perfluorinated iodine alkanes (PFCs), thereby resulting in environmental behaviors similar to those of FTOHs. Release of PFIs may increase the environmental burden of PFCs[1-2].
References
[1] Chang Wang. "Perfluorooctyl Iodide Stimulates Steroidogenesis in H295R Cells via a Cyclic Adenosine Monophosphate Signaling Pathway." Chemical Research in Toxicology 28 5 (2015): 848–854.
[2] WangYichen. "Estrogen-like response of perfluorooctyl iodide in male medaka (Oryzias latipes) based on hepatic vitellogenin induction." Environmental Toxicology 28 10 (2013): 571–8.
[3] Chang Wang. “Perfluorooctyl Iodide Stimulates Steroidogenesis in H295R Cells via a Cyclic Adenosine Monophosphate Signaling Pathway.” Chemical Research in Toxicology 28 5 (2015): 848–854.
[4] Maria Lumbierres, Adelina Vallribera, Marcial Moreno-Ma?as. “Addition of perfluorooctyl iodide to alkenes. Catalysis by triphenylphosphane.” Tetrahedron 58 20 (2002): Pages 4061-4065.
[5] X. Wang, Ai Fu, G. Chang. “Study on Preparation of 1H,1H,2H-Heptadecafluorodecene.” Advanced Materials Research 19 1 (2013): 2332–2335.
Properties of Perfluorooctyl iodide
| Melting point: | 25 °C |
| Boiling point: | 160-161 °C(lit.) |
| Density | 2.067 g/mL at 20 °C(lit.) |
| refractive index | n |
| Flash point: | 160-161°C |
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
| solubility | Chloroform, Ethyl Acetate (Slightly), Methanol (Slightly) |
| form | Liquid After Melting |
| color | Clear colorless to yellow or light pink |
| Sensitive | Light Sensitive |
| BRN | 1717141 |
| Exposure limits | ACGIH: TWA 0.01 ppm |
| CAS DataBase Reference | 507-63-1(CAS DataBase Reference) |
| NIST Chemistry Reference | Perfluorooctyl iodide(507-63-1) |
| EPA Substance Registry System | Octane, 1,1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8-heptadecafluoro-8-iodo- (507-63-1) |
Safety information for Perfluorooctyl iodide
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Perfluorooctyl iodide
| InChIKey | KWXGJTSJUKTDQU-UHFFFAOYSA-N |
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran

You may like
-
 Perfluorooctyl iodide 98% CAS 507-63-1View Details
Perfluorooctyl iodide 98% CAS 507-63-1View Details
507-63-1 -
 Heptadecafluoro-n-octyl Iodide CAS 507-63-1View Details
Heptadecafluoro-n-octyl Iodide CAS 507-63-1View Details
507-63-1 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 614-19-7 98%View Details
614-19-7 98%View Details
614-19-7 -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 -
 4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
5305-40-8
