OXAMNIQUINE(200MG)DISCONTINUED
- CAS NO.:21738-42-1
- Empirical Formula: C14H21N3O3
- Molecular Weight: 279.33484
- MDL number: MFCD00866725
- EINECS: 2445564
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-10-23 13:36:13
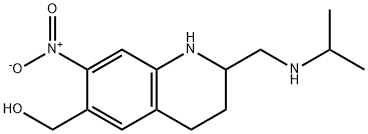
What is OXAMNIQUINE(200MG)DISCONTINUED?
Absorption
Well absorbed orally
Description
Oxamniquine was originally investigated in the 1960s and was found to have limited antiprotozoal activity, with activity against Schi stosoma mansoni but no activity against the other two schistosomal organisms. In addition, the drug is stage specific, with activity against cercariae and very young schistosomula and adult worms. For reasons that remain unknown, the drug is more effective against adult male worms than against female worms. The drug has structural similarity to hycanthone, which is no longer used because of severe toxicity and teratogenic effects.
Originator
Vansil, Pfizer , US ,1980
The Uses of OXAMNIQUINE(200MG)DISCONTINUED
Antischistosomal.
Background
An anthelmintic with schistosomicidal activity against Schistosoma mansoni, but not against other Schistosoma spp. Oxamniquine causes worms to shift from the mesenteric veins to the liver where the male worms are retained; the female worms return to the mesentery, but can no longer release eggs. (From Martidale, The Extra Pharmacopoeia, 31st ed, p121)
Indications
For treatment of Schistosomiasis caused by Schistosoma mansoni
Definition
ChEBI: A member of the class of quinolines that is 1,2,3,4-tetrahydroquinoline which is substituted at positions 2, 6, and 7 by (isopropylamino)methyl, hydroxymethyl, and nitro groups, respectively.
Indications
Oxamniquine (Vansil) is a tetrahydroquinoline that
stimulates parasite muscular activity at low concentrations
but causes paralysis at higher concentrations. The
drug may act by esterification and binding of DNA,
leading to the death of the schistosome by interruption
of its nucleic acid and protein synthesis. The fluke may
esterify oxamniquine to produce a reactive metabolite
that alkylates parasite DNA. Resistance results from
absent or defective esterifying activity of the drug.
Oxamniquine has a restricted range of efficacy, being
active only against S. mansoni infections.
Oxamniquine is given orally and is readily absorbed
from the intestinal tract. Peak concentrations in plasma
are obtained in about 3 hours. The drug is excreted in
urine mostly as a 6-carboxyl derivative.
Side effects include CNS toxicity with unsteadiness
and occasionally seizures, especially in patients with a
history of seizures. It is contraindicated in pregnancy.
Manufacturing Process
(1) Four fermenters are set up, each one of which contained 2.0 liters of the
following medium, sterilized for 35 minutes at 15 psi, respectively
The fermenters are inoculated with 7.5% by volume of a 24-hour old culture of Aspergillus sclerotiorum Huber grown at 28°C in 50 ml aliquots of the above described soybean-glucose medium contained in 300 ml Erlenmeyer flasks, placed on a shaker rotating at approximately 230 rpm. The inoculated fermenters are agitated at 1,380 rpm and each aerated with 1 liter of air per minute and at a temperature of 28°C for 47 hours. A silicone antifoam is added when required. At the end of the 47 hour period, the pH of the fermentation broth rose to 6.8 to 6.9. Sulfuric acid is then added with sterile precautions to restore the pH to 6.5.
(2) 0.75 g of 2-isopropylaminomethyl-6-methyl-7-nitro-1,2,3,4tetrahydroquinoline as hydrogen maleate, dissolved in 75 ml of sterile water, is added to each of the four fermenters and agitation and aeration are continued for a further 23 hours. The whole fermentation broths from each fermenter are pooled, the pH adjusted to 8.0 with sodium hydroxide and the 8.2 liters of fermentation broth thus obtained are extracted by agitating vigorously with 16.4 liters of methylene chloride for 10 minutes. The solvent extract is then dried over anhydrous sodium sulfate and subsequently evaporated to dryness at a temperature below 40°C (dry weight 5.567 g).
(3) The dark brown residue from (2) is extracted four times with methanol at room temperature, decanting the solution from the insoluble material. The combined methanol extracts, total volume about 200 ml, are then filtered and treated with 3 g of sodium borohydride, added in portions over a period of 30 minutes with stirring, to reduce any 6-formyl compound present to the 6hydroxymethyl compound. The methanol solution is then allowed to stand overnight at room temperature and is thereafter diluted with 1 liter of ether. The solution is washed 4 times with 500 ml of water and the resulting pale yellow ethereal solution is dried over magnesium sulfate. The ether is next removed by vacuum distillation from a water bath at 40°C. The residue is dissolved in about 75 ml of isopropanol at 50°C, filtered to remove any insoluble particles and cooled overnight in the refrigerator. The product is collected and dried in vacuo to yield 0.5 g of 6-hydroxymethyl-2isopropylaminomethyl-7-nitro-1,2,3,4-tetrahydroquinoline as pale yellow crystals of melting point 147°C to 149°C. A further 0.5 g of crude material is obtained from the mother liquors of the recrystallization. Total yield is therefore 1.0 g (0.0036 mol) from 3.0 g (0.0079 mol) of starting material, i.e., 45% of the theoretical amount.
brand name
Vansil (Pfizer).
Therapeutic Function
Antischistosomal
Antimicrobial activity
Activity is restricted to Schistosoma mansoni. Some strains, particularly those in Egypt and Southern Africa, require higher doses for efficacy owing to innate tolerance.
Pharmaceutical Applications
A synthetic quinolinemethanol, available for oral administration.
Mechanism of action
Oxamniquine is activated via esterification to a biological ester that spontaneously dissociates to an electrophile, which alkylates the helminth DNA, leading to irreversible inhibition of nucleic acid metabolism. Resistant helminths do not esterify oxamniquine; therefore, activation does not occur. Other metabolic reactions consist of oxidative reactions, leading to inactivation. The metabolites are excreted primarily in the urine.
Pharmacokinetics
Oxamniquine is an anthelmintic with schistosomicidal activity against Schistosoma mansoni, but not against other Schistosoma spp. Oxamniquine causes worms to shift from the mesenteric veins to the liver where the male worms are retained; the female worms return to the mesentery, but can no longer release egg.
Pharmacokinetics
It is rapidly absorbed after oral administration, achieving a peak concentration of 0.3–2.5 mg/L 1–3 h after an oral dose of 15 mg/kg body weight. Peak levels following intramuscular treatment at 7.5 mg/kg generally do not exceed 0.15 mg/L. It is extensively metabolized to biologically inactive 6-carboxylic and 2-carboxylic acid derivatives, which are excreted in the urine, mostly within 12 h.
Clinical Use
Infection with S. mansoni
Clinical Use
1,2,3,4-Tetrahydro-2-[(isopropylamino)methyl]-7-nitro-6-quinolinemethanol (Vansil) is an antischistosomal agent thatis indicated for the treatment of Schistosoma mansoni (intestinalschistosomiasis) infection. It has been shown to inhibitDNA, RNA, and protein synthesis in schistosomes. The6-hydroxymethyl group is critical for activity; metabolic activationof precursor 6-methyl derivatives is critical. Theoral bioavailability of oxamniquine is good; effectiveplasma levels are achieved in 1 to 1.5 hours. The plasmahalf-life is 1 to 2.5 hours. The drug is extensively metabolizedto inactive metabolites, of which the principal one isthe 6-carboxy derivative.
The free base occurs as a yellow crystalline solid thatis slightly soluble in water but soluble in dilute aqueousmineral acids and soluble in most organic solvents. Itis available in capsules containing 250 mg of the drug.Oxamniquine is generally well tolerated. Dizziness anddrowsiness are common, but transitory, side effects. Seriousreactions, such as epileptiform convulsions, are rare.
Side Effects
Dizziness, sleepiness, nausea and headache occur frequently. Other side effects are probably due to the death and disintegration of the worms in the liver. Following treatment, urine may become red.
Safety Profile
Poison by ingestion, intraperitoneal and intramuscular routes. Human mutation data reported. An antischistosomal agent. When heated to decomposition it emits toxic fumes of NOx.
Metabolism
Probably hepatic
Properties of OXAMNIQUINE(200MG)DISCONTINUED
| Melting point: | 147-149° |
| Boiling point: | 422.1°C (rough estimate) |
| Density | 1.1119 (rough estimate) |
| refractive index | 1.5700 (estimate) |
| solubility | DMSO (Slightly), Methanol (Slightly) |
| pka | pKa 3.28 ± 0.07;9.53(H2O,t =25) (Uncertain) |
| form | Solid |
| color | Light-orange powder |
| Stability: | Hygroscopic |
Safety information for OXAMNIQUINE(200MG)DISCONTINUED
Computed Descriptors for OXAMNIQUINE(200MG)DISCONTINUED
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 3,4-Dibenzyloxybenzaldehyde 4-Hydrazinobenzoic acid 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 3-NITRO-2-METHYL ANILINE 4-IODO BENZOIC ACID 4-HYDROXY BENZYL ALCOHOL (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 4-methoxy-3,5-dinitropyridine 2-(Cyanocyclohexyl)acetic acid 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride tert-butyl 4- (ureidomethyl)benzylcarbamate diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 (9H-fluoren-9-yl)methyl (2,5-dioxopyrrolidin-1-yl) carbonate 82911-69-1 98.0%View Details
(9H-fluoren-9-yl)methyl (2,5-dioxopyrrolidin-1-yl) carbonate 82911-69-1 98.0%View Details
82911-69-1 -
 13057-17-5 95.0%View Details
13057-17-5 95.0%View Details
13057-17-5 -
![2-Nitro-8,9-dihydro-5H-benzo [7] annulen-7(6H)-one 98.0%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-Nitro-8,9-dihydro-5H-benzo [7] annulen-7(6H)-one 98.0%View Details
2-Nitro-8,9-dihydro-5H-benzo [7] annulen-7(6H)-one 98.0%View Details
740842-50-6 -
 4-bromoaniline 106-40-1 99.0%View Details
4-bromoaniline 106-40-1 99.0%View Details
106-40-1 -
 1421517-99-8 99.0%View Details
1421517-99-8 99.0%View Details
1421517-99-8 -
 5-bromo-2-chlorobenzoic acid 99.0%View Details
5-bromo-2-chlorobenzoic acid 99.0%View Details
21739-92-4 -
 2-methyl-5-nitrophenol 98.0%View Details
2-methyl-5-nitrophenol 98.0%View Details
5428-54-6 -
 15761-38-3 97.0%View Details
15761-38-3 97.0%View Details
15761-38-3