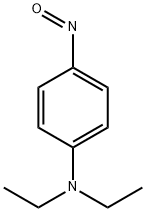
N,N-Dimethylaniline
Synonym(s):Dimethylaminobenzene;N,N-Dimethylaniline
- CAS NO.:121-69-7
- Empirical Formula: C8H11N
- Molecular Weight: 121.18
- MDL number: MFCD00008304
- EINECS: 204-493-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-01-27 09:38:02
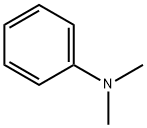
What is N,N-Dimethylaniline?
Description
DMA, an aromatic amine, is a straw-coloredliquid with a characteristic amine-like odor. It turns brownon contact with air. Molecular weight= 121.20; Boilingpoint= 192℃; Freezing/Melting point = 2.2℃; Vaporpressure = 1 mmHg at 20℃; Flash point = 61℃;Autoignition temperature= 371℃. Explosive limits:LEL = 1%; UEL= 7%. Insoluble in water.
Chemical properties
N,N-Dimethylaniline is a light yellow to light brown oily liquid. Has a pungent odor. Soluble in ethanol, chloroform, ether and aromatic organic solvents, slightly soluble in water.It is a tertiary amine used in the synthesis of several triarylmethane dyes like malachite green. It is also used in the synthesis of a magnetic gram stain for the detection of bacteria.
Physical properties
Straw to brown-colored oily liquid with a characteristic amine-like odor. Odor threshold concentration is 13 ppb (quoted, Amoore and Hautala, 1983).
The Uses of N,N-Dimethylaniline
N,N-Dimethylaniline is used in production of dyestuffs, as a solvent, a reagent in methylation reactions, and a hardener in fiberglass reinforced resins.
The Uses of N,N-Dimethylaniline
N,N-dimethylaniline is an important dye intermediate. It can be used to prepare alkaline yellow, Crystal Violet 5BN, basic magenta green, basic lake blue BB, basic brilliant blue R, cationic red 2BL, brilliant red 5GN, violet 3BL, brilliant blue, etc. In the pharmaceutical industry, the product can be used to manufacture cefazolin V, sulfamonomethoxine, Sulfadoxine, fluorocytosine, etc. moreover, It can be used as an intermediate to vanillin, a stabilizer for colorimetric peroxidase determination or as a reagent in chemical synthesis.
Definition
ChEBI: N,N-dimethylaniline is a tertiary amine that is aniline in which the amino hydrogens are replaced by two methyl groups. It is a tertiary amine and a dimethylaniline.
Production Methods
N,N-Dimethylaniline is made by heating aniline, methyl alcohol and sulfuric acid under pressure, followed by hydrolysis of the sulfate formed with sodium hydroxide to the free base (Windholz et al 1983). United States production in 1975 was estimated at 4,600 metric tons (HSDB 1989).
General Description
N,N-Dimethylaniline appears as a yellow to brown colored oily liquid with a fishlike odor. Less dense than water and insoluble in water. Flash point 150°F. Toxic by ingestion, inhalation, and skin absorption. Used to make dyes and as a solvent.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
Explosive decomposition occurred when finely divided benzoyl peroxide was allowed to react with N,N-Dimethylaniline by breaking an ampoule containing 0.5 grams of dimethylaniline in an autoclave, NFPA 491M, 1991. This result may be expected with other peroxides and various oxidants.
Health Hazard
TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Health Hazard
Clinical signs of intoxication with N,N-dimethylaniline in man are headaches, cyanosis, dizziness, labored breathing, paralysis and convulsions (HSDB 1989). It is absorbed through the skin to produce a dangerous methemoglobinemia (Gosselin 1984). Treatment is similar to that of aniline with the object of managing methemoglobinemia.
Fire Hazard
Combustible material: may burn but does not ignite readily. When heated, vapors may form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated. Runoff may pollute waterways. Substance may be transported in a molten form.
Flammability and Explosibility
Non flammable
Industrial uses
N,N-Dimethylaniline is used as chemical intermediate in the manufacturing of vanillin, Michler's ketone, and dyes such as Acid Red 2, Basic Green 4 and Basic Violet 1 (Northcott 1978). It is also used as a solvent and an activator for polyesters and as an alkylating agent (Beard and Noe 1981). It is used as an acid scavenger or accepter in the manufacture of beta-lactam antibiotics such as penicillin and cephalosporin (Nachtmann and Gstrein 1981).
Safety Profile
Suspected carcinogen with equivocal tumorigenic data. Human poison by ingestion. Moderately toxic by inhalation and skin contact. A skin irritant. Human systemic effects by ingestion: nausea or vomiting. Physiological action is similar to, but less toxic than, adne. A central nervous system depressant. Mutation data reported. Flammable liquid when exposed to heat, flame, or oxidizers. Explodes on contact with benzoyl peroxide or disopropyl peroxydicarbonate. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits htghly toxic fumes of adne and NOx. See also ANILINE.
Potential Exposure
Tumorigen,Mutagen, Human Data; Primary Irritant. This material isused as a chemical intermediate in the manufacture of manydyes and rubber chemicals; solvent, emulsifier, and analytical reagent.
Carcinogenicity
A 2-year corn oil gavage bioassay conducted by NTP in F344/N rats (0.3 or 30 mg/kg) and B6C3F1 mice (0, 15, or 30 mg/kg) for 103 weeks concluded that there was some evidence of carcinogenic activity for male F344/N rats as indicated by the increased incidences of sarcomas or osteosarcomas in the spleen; there was no evidence of carcinogenic activity in the female rats or male mice; there was equivocal evidence of carcinogenic activity for female mice as indicated by an increased incidence of squamous cell papillomas of the forestomach. Both rats and mice could have tolerated doses higher than those used in these studies.
Environmental Fate
Photolytic. A rate constant of 1.48 x 10-10 cm3/molecule?sec was reported for the reaction of
N,N-dimethylaniline and OH radicals in air at room temperature (Atkinson et al., 1987).
Chemical/Physical. Products identified from the gas-phase reaction of ozone with N,Ndimethylaniline
in synthetic air at 23 °C were: N-methylformanilide, formaldehyde, formic acid,
hydrogen peroxide, and a nitrated salt having the formula: [C6H6NH(CH3)2]+NO3
- (Atkinson et al.,
1987). Reacts with acids forming water-soluble salts.
Metabolism
Ν,Ν-Dimethylaniline undergoes N-demethylation, N-oxidation, and ring hydroxylation in animals. Urinary metabolites produced by dogs and rabbits injected with this compound included 4-aminophenol, 4-dimethylaminophenol, 2-aminophenol and N-methylaniline (Williams 1959; Kiese and Renner 1974). N-oxidation, N-demethylation and ring hydroxylation of Ν,Ν-dimethylaniline was demonstrated in vitro using liver microsomal preparations from pigs, rats, rabbits, chickens, and guinea pigs (Fish et al 1955; Zeigler and Pettit 1964, 1966; Abou-Donia and Menzel 1968). N-oxidation was also demonstrated using whole homogenate of human liver (Zeigler and Gold 1971; Rane 1974). Evidence has been obtained with rabbit liver microsomes for two pathways for Ν,Ν-dimethylaniline N-oxidation (Hlavica and Kehl 1977). One pathway involves cytochrome P-450 while the second makes use of flavin-containing monooxygenase. N,N-Dimethylaniline was also metabolized to formaldehyde in the nasal and respiratory mucosa of Fischer 344 rats (McNulty et al 1983). Alveolar type II cells from rabbit and rat lungs catalyzed the N-oxidation of this compound (Devereux and Fouts 1974; Ohmiya and Mehendale 1981). The microsomal preparation from rat seminal vesicles when fortified with arachidonic acid catalyzed the dealkylation of N,Ndimethylaniline (Sivarajah et al 1982).
Shipping
This compound requires a shipping label of“POISONOUS/TOXIC MATERIALS.” It falls in HazardClass 6.1 and Packing Group II.
Purification Methods
Primary and secondary amines (including aniline and monomethylaniline) can be removed by refluxing for 4-5hours with excess acetic anhydride, and then fractionally distilling. Crocker and Jones (J Chem Soc 1808 1959) used four volumes of acetic anhydride, then distilled off the greater part of it, and dissolved the residue in ice-cold dilute HCl. Non-basic materials were removed by ether extraction, then the dimethylaniline was liberated with ammonia, extracted with ether, dried, and distilled under reduced pressure. Metzler and Tobolsky (J Am Chem Soc 76 5178 1954) refluxed with only 10% (w/w) of acetic anhydride, then cooled and poured it into excess 20% HCl, which, after cooling, was extracted with diethyl ether. (The amine hydrochloride remains in the aqueous phase.) The HCl solution was cautiously made alkaline to phenolphthalein, and the amine layer was drawn off, dried over KOH and fractionally distilled under reduced pressure, under nitrogen. Suitable drying agents for dimethylaniline include NaOH, BaO, CaSO4, and CaH2. Other purification procedures include the formation of the picrate (m 163o from Me2CO or EtOH/H2O), prepared in *benzene solution and crystallised to constant melting point, then decomposed with warm 10% NaOH and extracted into ether: the extract was washed with water and distilled under reduced pressure. The oxalate salt has also been used for purification. The base has been fractionally crystallised by partial freezing and also from aqueous 80% EtOH then from absolute EtOH. It has been distilled from zinc dust, under nitrogen. [Beilstein 12 H 141, 12 I 151, 12 II 2, 12 III 245, 12 IV 243.]
Incompatibilities
Forms explosive mixture with air.Contact with strong oxidizers, strong acids, benzoyl peroxide may cause fire and explosion hazard. Contact withhypochlorite bleaches form explosive chloramines.Incompatible with anhydrides, isocyanates, aldehydes.
References
N,N-Dimethylaniline and 1-(trifluoromethyl)benzene-functionalized tetrakis(ethynyl)pyrenes: synthesis, photophysical, electrochemical and computational studies DOI:10.1016/J.TET.2011.12.066
Friedel-Crafts Reaction of N,N-Dimethylaniline with Alkenes Catalyzed by Cyclic Diaminocarbene-Gold(I) Complex doi: 10.1038/s41598-018-29854-0
Groundwater Chemicals Desk Reference DOI:10.5860/choice.27-6127
Patty's Toxicology 6-Volume Set-Wiley (2012)
Proctor Proctor and Hughes' chemical hazards of the workplace (5th edn) Wiley Interscience DOI:10.1002/JAT.1034
Nitrogen and phosphorus solvents DOI:10.1016/c2009-0-01359-2
Purification of Laboratory Chemicals DOI:10.5860/choice.50-6768
Properties of N,N-Dimethylaniline
| Melting point: | 1.5-2.5 °C (lit.) |
| Boiling point: | 193-194 °C (lit.) |
| Density | 0.956 g/mL at 25 °C (lit.) |
| vapor density | 3 (vs air) |
| vapor pressure | 2 mm Hg ( 25 °C) |
| refractive index | n |
| Flash point: | 158 °F |
| storage temp. | Store below +30°C. |
| solubility | 1.2g/l |
| form | Liquid |
| pka | 5.15(at 25℃) |
| color | Clear yellow |
| Relative polarity | 0.179 |
| PH | 7.4 (1.2g/l, H2O, 20℃) |
| explosive limit | 1.2-7%(V) |
| Water Solubility | 1 g/L (20 ºC) |
| Merck | 14,3234 |
| BRN | 507140 |
| Henry's Law Constant | 4.98(x 10-6 atm?m3/mol) at 20 °C (approximate - calculated from water solubility and vapor pressure) |
| Exposure limits | NIOSH REL: TWA 5 ppm (25 mg/m3), STEL 10 ppm (50 mg/m3), IDLH 100
ppm; OSHA PEL: TWA 5 ppm; ACGIH TLV: TWA 5 ppm, STEL 10 ppm (adopted). |
| Dielectric constant | 4.4000000000000004 |
| Stability: | Stable. Incompatible with strong oxidizing agents, strong acids, acid chlorides, acid anhydrides, chloroformates, halogens. Combustible. |
| CAS DataBase Reference | 121-69-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Benzenamine, N,N-dimethyl-(121-69-7) |
| IARC | 3 (Vol. 57) 1993 |
| EPA Substance Registry System | N,N-Dimethylaniline (121-69-7) |
Safety information for N,N-Dimethylaniline
| Signal word | Danger |
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06  Health Hazard GHS08  Environment GHS09 |
| GHS Hazard Statements |
H351:Carcinogenicity H411:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P201:Obtain special instructions before use. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P310:IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician. |
Computed Descriptors for N,N-Dimethylaniline
| InChIKey | JLTDJTHDQAWBAV-UHFFFAOYSA-N |
N,N-Dimethylaniline manufacturer
ARRAKIS INDUSTRIES LLP
ASM Organics
New Products
4-Piperidinemethanol Ethyl 2,4-Dihydroxy-6-methylnicotinate Ethyl isonicotinate 3-pyridine methanol N-Methyl 4-chloro-pyridine-2-carboxamide 2-Fluoro-6-iodobenzoic acid 2-((2,6-difluorobenzyl)(ethoxycarbonyl)amino)-4-((dimethylamino)methyl)-5-(4-nitrophenyl)thiophene-3-carboxylic acid Ethyl2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate Elinzanetant tert-butyl 2-(4-amino-6-chloropyrimidin-5-yloxy)ethylmethylcarbamate Phenylazomalononitrile 5,6 Dimethoxy-1-indanone 3-Iodophenylacetic acid 2-Hexyn-1-ol Dibenzo-18-crown-6 2-Propanamine, 1-chloro-, hydrochloride (9CI) 3-Pyridineacetonitrile, α-hydroxy- 3-(hexyloxy)-4-(pyridin-3-yl)-1,2,5-thiadiazole N Ethylmethylamine Ethyl Methanesulfonate N N' DimethylEthylenediamine Lead II Bromide Variamine Blue B Diazonium salt N N N'Trimethyl ethylenediamineRelated products of tetrahydrofuran
You may like
-
![N,N-Dimethylaniline [for Biochemical Research] CAS 121-69-7](https://img.chemicalbook.in//Content/image/CP5.jpg) N,N-Dimethylaniline [for Biochemical Research] CAS 121-69-7View Details
N,N-Dimethylaniline [for Biochemical Research] CAS 121-69-7View Details
121-69-7 -
 N,N-Dimethylaniline, GR 99%+ CAS 121-69-7View Details
N,N-Dimethylaniline, GR 99%+ CAS 121-69-7View Details
121-69-7 -
 N: N Dimethyl Aniline (SQ) CAS 121-69-7View Details
N: N Dimethyl Aniline (SQ) CAS 121-69-7View Details
121-69-7 -
 N, N-Dimethylaniline CAS 121-69-7View Details
N, N-Dimethylaniline CAS 121-69-7View Details
121-69-7 -
 N,N-Dimethylaniline CAS 121-69-7View Details
N,N-Dimethylaniline CAS 121-69-7View Details
121-69-7 -
 N N Dimethylaniline CASView Details
N N Dimethylaniline CASView Details -
 N,N-Dimethylaniline CAS 121-69-7View Details
N,N-Dimethylaniline CAS 121-69-7View Details
121-69-7 -
 N-N-Dimethylaniline CAS 121-69-7View Details
N-N-Dimethylaniline CAS 121-69-7View Details
121-69-7
