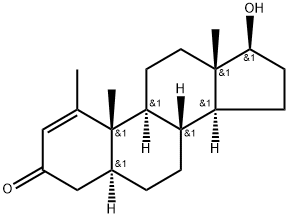
Methenolone acetate
- CAS NO.:434-05-9
- Empirical Formula: C22H32O3
- Molecular Weight: 344.49
- MDL number: MFCD00864181
- EINECS: 207-097-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-03-20 19:16:41
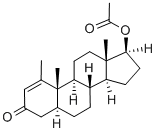
What is Methenolone acetate?
Description
Methenolone acetate (Brand name: Primobolan) is a synthetic, orally active anabolic-androgenic steroid and dihydrotestosterone (DHT) derivative. It can be used for the treatment of bone marrow disease and anemia. Methenolone acetate is known for its higher therapeutic efficiency and lower hepatic toxicity compared with its 17 alpha- alkylated analogs. However, it is frequently abused in human sports because of its capability of increasing muscle strength as well as promoting performance and aggressiveness. It is capable of enhancing performance in racehorses.
Chemical properties
White Solid
Originator
Primobolan ,Schering ,W. Germany,1961
The Uses of Methenolone acetate
Methenolone acetate is an anabolic steroid. This is a controlled substance.
Definition
ChEBI: Methenolone acetate is a steroid ester.
Manufacturing Process
8.42 ml of methyl iodide are slowly added dropwise at room temperature with
stirring in a nitrogen atmosphere to 3.067 g of magnesium turnings and 107
ml of absolute ether. After about 30 minutes, 185 ml of absolute
tetrahydrofuran are slowly introduced and then liquid is distilled off until a
boiling point of 62°C is reached. After cooling to room temperature, 613 mg
of cuprous chloride are added and then 10 g of ?1,4,6-androstatrien-17β-ol-3-
one-17-acetate in 110 ml of tetrahydrofuran slowly introduced. After 30
minutes reaction time, the whole is cooled to 0C, the excess of Grignard
reagent decomposed with saturated ammonium chloride solution, the product
diluted with ether and the aqueous phase separated. The ethereal phase is
washed consecutively with aqueous sodium thiosulfate solution, saturated
ammonium chloride solution and water. It is dried over sodium sulfate and
evaporated to dryness under vacuum. The residue is dissolved in 40 ml of
pyridine and 20 ml of acetic anhydride and the solution kept for 16 hours at
room temperature. It is then stirred into ice water and the precipitate filtered
with suction, dried and recrystallized from isopropyl ether. 1α-Methyl-?4,6-
androstadien-17β-ol-3-one-17-acetate is obtained. MP 156°C to 157°C; [α]D25
= -33.8° (in CHCl3; c = 0.9). Yield 65-70% of the theoretical.
4.67 g of 1α-methyl-?4,6-androstadien-17β-ol-3-one-17-acetate are dissolved
in 273 ml of methanol and, after the addition of 350 mg of 10% palladium on
calcium carbonate catalyst, hydrogenated until 1 mol equivalent of hydrogen
has been taken up. After filtering off the catalyst, the solution is treated with
150 ml of 2N-hydrochloric acid and evaporated under vacuum to about 1/3 of
the volume. The whole is then diluted with water and extracted with ether.
The ethereal solution is washed with water until neutral, dried over sodium
sulfate and evaporated. The crude product is heated on a steam bath for 90
minutes in 10 ml of pyridine and 10 ml of acetic anhydride. Extraction with
ether is then carried out and the ethereal phase washed until neutral with
water. The crude crystalline 1α-methyl-?4-androsten-17β-ol-3-one-17-
acetateobtained after drying and evaporation of the solution, melts at 122°C
to 129°C. Yield 98% of the theoretical.
1α-Methyl-?4-androsten-17β-ol-3-one-17-acetate when purified by
recrystallization from isopropyl ether melts at 138°C to 139°C.
Therapeutic Function
17β-Hydroxy-1β-methyl-5α-androst-l-ene-3-one acetate
References
Ho, Emmie N. M., et al. "Metabolic studies of methenolone acetate in horses." Analytica Chimica Acta 540.1(2005): 111-119.
https://en.wikipedia.org/wiki/Metenolone_acetate
Properties of Methenolone acetate
| Melting point: | 138-139° |
| Boiling point: | 419.57°C (rough estimate) |
| Density | 1.0906 (rough estimate) |
| refractive index | 1.5000 (estimate) |
| storage temp. | Store at -20°C |
| solubility | DMSO : ≥ 3.6 mg/mL (10.45 mM) |
| form | Solid |
| color | White to Off-White |
| InChI | InChI=1/C22H32O3/c1-13-11-16(24)12-15-5-6-17-18-7-8-20(25-14(2)23)21(18,3)10-9-19(17)22(13,15)4/h11,15,17-20H,5-10,12H2,1-4H3/t15-,17-,18-,19-,20-,21-,22-/s3 |
| CAS DataBase Reference | 434-05-9(CAS DataBase Reference) |
Safety information for Methenolone acetate
Computed Descriptors for Methenolone acetate
| InChIKey | PGAUJQOPTMSERF-MQEWATRANA-N |
| SMILES | [C@]12([H])CC[C@]3(C)[C@]([H])(CC[C@@]3([H])[C@]1([H])CC[C@@]1([H])CC(=O)C=C(C)[C@]21C)OC(=O)C |&1:0,4,6,10,12,16,24,r| |
Methenolone acetate manufacturer
New Products
3-Iodophenylacetic acid 3-Pyridineacetonitrile, α-hydroxy- 2-Propanamine, 1-chloro-, hydrochloride (9CI) 3-(hexyloxy)-4-(pyridin-3-yl)-1,2,5-thiadiazole 2-Hexyn-1-ol Dibenzo-18-crown-6 Nickel(II) perchlorate hexahydrate, 98% 4-Bromophenylacetonitrile, 95% 3-Bromo-4-fluoroaniline, 97% Sodium tetraborate decahydrate, 98% Palladium(II) acetate, trimer, Pd 99% 4-Bromo-2-chlorotoluene, 97% N N Dimethylformamide Dimethyl Acetal (Dmf Dma) 2,3-Dichloro Benzoyl Cyanide [Side Chain] Bis(2-Chloroethyl) Amine Hydrochloride L-Glutamic Acid Diethyl Ester Hydrochloride 5-(Difluoromethoxy)-2-Mercaptobenzimidazole 1-Ethyl-3-(3-Dimethylaminopropyl)-Carbodiimide Hydrochloride [EDC Hcl] 1,4-Napthoquinone Bromoiodomethane Sodium Bicarbonate Methylene Dichloride (MDC) Ethyl Acetate Indole-3-Carbinol (I3C)Related products of tetrahydrofuran

You may like
-
 17604-74-9 3-Pyridineacetonitrile, α-hydroxy- 98+View Details
17604-74-9 3-Pyridineacetonitrile, α-hydroxy- 98+View Details
17604-74-9 -
 131987-69-4 98+View Details
131987-69-4 98+View Details
131987-69-4 -
 2-Hexyn-1-ol 98+View Details
2-Hexyn-1-ol 98+View Details
764-60-3 -
 Cyclohexane, (2-propynyloxy)- 67967-07-1 98+View Details
Cyclohexane, (2-propynyloxy)- 67967-07-1 98+View Details
67967-07-1 -
 764-60-3 2-Hexyn-1-ol 98+View Details
764-60-3 2-Hexyn-1-ol 98+View Details
764-60-3 -
 2-Propanamine, 1-chloro-, hydrochloride (9CI) 98+View Details
2-Propanamine, 1-chloro-, hydrochloride (9CI) 98+View Details
5968-21-8 -
 3-Iodophenylacetic acid 1878-69-9 98+View Details
3-Iodophenylacetic acid 1878-69-9 98+View Details
1878-69-9 -
 132945-75-6 (S)-1-Boc-3-methanesulfonyloxy-pyrrolidine 98+View Details
132945-75-6 (S)-1-Boc-3-methanesulfonyloxy-pyrrolidine 98+View Details
132945-75-6
