INACTIN
Synonym(s):Thiobutabarbital sodium salt hydrate
- CAS NO.:947-08-0
- Empirical Formula: C10H15N2NaO2S
- Molecular Weight: 250.29
- MDL number: MFCD00214068
- EINECS: 213-425-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-03-19 15:37:50
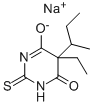
What is INACTIN?
Description
Thiobutabarbital sodium , the sodium salt of 5-sec-butyl-5-ethylthiobarbituric acid, is a yellowishwhite amorphous powder soluble in water and alcohol. The colorless crystals of the free acid melt at 163 – 165 ?C. Thiobutabarbital is prepared from thiourea and a malonic ester .
The Uses of INACTIN
Inactin? hydrate has been used to anesthetize rats to study stress-induced hypersensitivity and mice to study Cisplatin-induced neuropathy. It has also been used to anesthetize rats to measure the glomerular filtration rate (GFR).
General Description
Crystals.
Air & Water Reactions
Slight hygroscopic. Water soluble.
Reactivity Profile
An amine, organosulfide. Organosulfides are incompatible with acids, diazo and azo compounds, halocarbons, isocyanates, aldehydes, alkali metals, nitrides, hydrides, and other strong reducing agents. Reactions with these materials generate heat and in many cases hydrogen gas. Many of these compounds may liberate hydrogen sulfide upon decomposition or reaction with an acid. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides.
Fire Hazard
Flash point data for INACTIN are not available. INACTIN is probably combustible.
Biochem/physiol Actions
Inactin? is a long-lasting rodent anesthetic with minimal effects on cardiovascular tone and renal output. It exhibits sedative and hypnotic properties.
Clinical Use
It is a short-acting intravenous anesthetic. It is the least potent of the thiobarbiturates and therefore causes the least number of complications during anesthesia. The compound is available as 5 % and 10 % solutions.
Properties of INACTIN
| Melting point: | 167-168 °C |
| solubility | H2O: storage of solutions for more than 8 hours at 4°C is not recommended.soluble |
| form | solid |
| color | light yellow |
| PH | pH:9.0~11.0 (50g/l, 25℃) |
| EPA Substance Registry System | Thiobutabarbital sodium (947-08-0) |
Safety information for INACTIN
Computed Descriptors for INACTIN
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran

![2-ethyl-2-[(methylamino)methyl]butan-1-ol](https://img.chemicalbook.in/CAS/GIF/959238-57-4.gif)
You may like
-
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 88491-46-7 98%View Details
88491-46-7 98%View Details
88491-46-7 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4