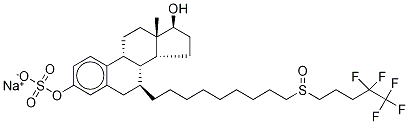
Fulvestrant
Synonym(s):(7α,17β)-7-[9-[(4,4,5,5,5-Pentafluoropentyl)sulfinyl]nonyl]estra-1,3,5(10)-triene-3,17-diol;Faslodex;Fulvestrant;
- CAS NO.:129453-61-8
- Empirical Formula: C32H47F5O3S
- Molecular Weight: 606.77
- MDL number: MFCD00903953
- EINECS: 642-998-6
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-26 08:49:36
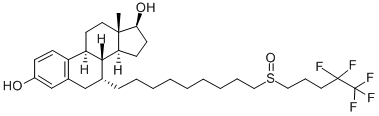
What is Fulvestrant?
Toxicity
There is no clinical experience with overdosage in humans.
Description
Fulvestrant was launched in the US as a novel once monthly injectable steroidal estrogen antagonist for the treatment of hormone receptor positive metastatic breast cancer in postmenopausal women with disease progression following estrogen therapy. This 7a-alkylsulphinyl derivative of estradiol can be prepared in 10 steps from 6,7- didehydro-19-nor-testosterone by successive conjugate addition of the organocuprate derived from O-protected 9-bromononan-l-o1 followed by aromatization of the resulting enone, then activation of the protected primary alcohol, substitution with 4,4,5,5,5- pentafluoropentanthiol and oxidation to the sulfoxide. Fulvestrant is the first “pure” estrogen antagonist from a novel class known as selective estrogen receptor down regulators (SERDs). It binds to the estrogen receptor (ER), with affinity close to that of estradiol and 100 fold greater than that of tamoxifen (a partial estrogen antagonist), preventing estrogen-stimulated gene activation, thereby interfering with the estrogenrelated processes essential for cell-cycle completion. Fulvestrant also appears to downregulate the ER by 80-90% often to non detectable level both in vitro and in vivo. In comparison to tamoxifen, fulvestrant is devoid of systemic estrogenic activity, it displays no uterotrophic activity and is able to block the uterine stimulation of estradiol or tamoxifen. Furthermore, fulvestrant completely blocks the cell growth in tamoxifen-resistant breast cancer cell-lines and prevents growth of tamoxifen resistant tumor in mice. In clinical trials, it was also shown that fulvestrant is comparable to anastrozole (a third generation aromatase inhibitor) both in efficacy and tolerability in postmenopausal women with tamoxifen-resistant advanced breast cancers.
Chemical properties
White or almost white powder.
Originator
Astra Zeneca (UK)
The Uses of Fulvestrant
A novel steroidal estrogen antagonist reported to lack any partial agonist activity. Antineoplastic (hormonal).
The Uses of Fulvestrant
antiestrogen
Indications
For the treatment of hormone receptor positive metastatic breast cancer in postmenopausal women with disease progression following anti-estrogen therapy, as monotherapy or in combination with other antineoplastic agents.
What are the applications of Application
ICI 182,780 is an estrogen receptor inhibitor and GPR30 activator
Background
Fulvestrant is a drug treatment of hormone receptor (HR)-positive metastatic breast cancer in post-menopausal women with disease progression following anti-estrogen therapy. It is an estrogen receptor antagonist with no agonist effects, which works both by down-regulating and by degrading the estrogen receptor. While it is used as monotherapy for the treatment of breast cancers, it is also used in combination with alpelisib for the treatment of HR-positive, human epidermal growth factor receptor 2 (HER2)-negative, PIK3CA-mutated, advanced or metastatic breast cancer
Definition
ChEBI: A 3-hydroxy steroid that is 17beta-estradiol in which the 7alpha hydrogen has been replaced by a nonyl group in which one of the hydrogens of the terminal methyl has been replaced by a (4,4,5,5,5-pentafluoropentyl)sulfinyl group. An estrogen receptor antagonist, it is used in the treatment of breast cancer.
brand name
Faslodex (AstraZeneca).
General Description
Fulvestrant, 7α-[9-[(4,4,5,5,5-pentafluoropentyl)sulfinyl]nonyl]estra-1,3,5(10)-triene-3,17β-diol (Faslodex), is an antagonist structurally based onthe estradiol structure, with a long, substituted alkyl chainattached at the 7α-position of the steroid skeleton. Whenbound to the ERs, this alkyl chain induces a conformationof the receptor distinctive from that formed upon estradiolor tamoxifen binding, preventing agonist action.Fulvestrant is a pure antagonist at both ERαand ERβandan ER downregulator (stimulates degradation of the ER),completely lacking the agonist activity that is seen with tamoxifenor raloxifene. The different pharmacological profileof fulvestrant allows the use of this agent in womenwho have had disease progression after prior antiestrogentherapy (typically tamoxifen), providing an alternative toaromatase inhibitors.
Biological Activity
A high affinity estrogen receptor antagonist (IC 50 = 0.29 nM), devoid of any partial agonism both in vitro and in vivo . Also high affinity agonist at the membrane estrogen receptor GPR30.
Biochem/physiol Actions
Fulvestrant (ICI 182,780) is a selective estrogen receptor down-regulator (SERD). Fulvestrant is a high affinity estrogen receptor antagonist. IC50 = 0.29 nM. Fulvestrant is the first "pure" antiestrogen with no agonistic activity both in vitro and in vivo.
Pharmacokinetics
Fulvestrant for intramuscular administration is an estrogen receptor antagonist without known agonist effects.
Clinical Use
Treatment of postmenopausal women with oestrogenreceptor- positive, locally advanced or metastatic breast cancer
Side Effects
Side effects appear to be minimal and include several GI symptoms , headache, and hot flashes . There is no clinical evidence of uterine stimulation or laboratory evidence of stimulation of endometrial carcinoma models. Fulvestrant should not be adm inistered to women who are pregnant, who are taking antic oagulants, or who have thrombocytopenia.
Synthesis
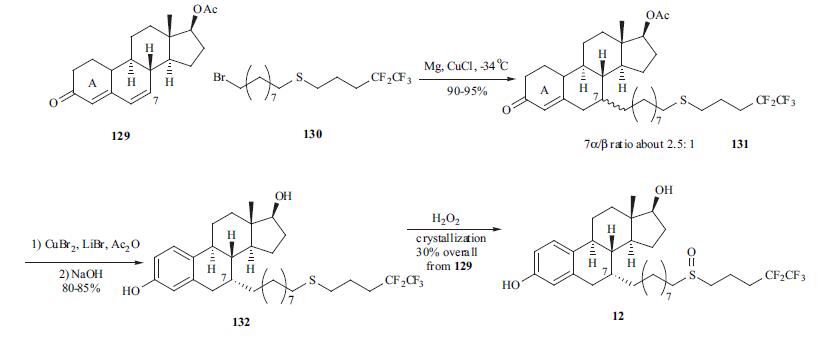
Fulvestrant is administered as a once a month i. m. injection. Several routes for the synthesis of fulvestrant (12) were published. One of the best routes is depicted in the scheme. The conjugate addition of Grignard reagent derived from bromide 130 with dienone 129 gave adduct 131 as a mixture of 7|á- and 7|?-isomers in a ratio of 2.5:1 in 90-95% yield. Aromatization of the A-ring with copper bromide/lithium bromide in acetic acid followed by hydrolysis of the ester group provided diol 132 in 80-85% yield. Oxidation of the side chain from sulfite to sulfone followed by crystallization provided fulvestrant (12) in 30% overall yield from dienone 129.
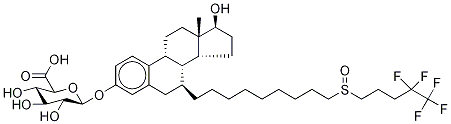
Metabolism
Metabolism of fulvestrant appears to involve combinations of a number of possible biotransformation pathways analogous to those of endogenous steroids, including oxidation, aromatic hydroxylation, conjugation with glucuronic acid and/or sulphate at the 2, 3 and 17 positions of the steroid nucleus, and oxidation of the side chain sulphoxide. Identified metabolites are either less active or exhibit similar activity to fulvestrant in antiestrogen models. Studies using human liver preparations and recombinant human enzymes indicate that cytochrome P-450 3A4 (CYP 3A4) is the only P-450 isoenzyme involved in the oxidation of fulvestrant; however, the relative contribution of P-450 and non-P-450 routes in vivo is unknown.
Metabolism
The metabolism of fulvestrant has not been fully
evaluated, but involves combinations of a number of
possible biotransformation pathways analogous to those
of endogenous steroids. Identified metabolites (includes
17-ketone, sulphone, 3-sulphate, 3- and 17-glucuronide
metabolites) are either less active or exhibit similar
activity to fulvestrant in anti-oestrogen models.
Fulvestrant is eliminated mainly in metabolised form. The
major route of excretion is via the faeces.
Storage
Room temperature
References
Osborne et al. (2004), Fulvestrant: an oestrogen receptor antagonist with a novel mechanism of action; Br. J. Cancer 90 (Suppl 1):S2 Thomas et al. (2005), Identity of an estrogen membrane receptor coupled to a G protein in human breast cancer cells; Endocrinology 146 624 Wardley (2002), Fulvestrant: a review of its development, pre-clinical and clinical data; Int. J. Clin. Pract. ?56 305 Castro et al. (2012),?Coumestrol has neuroprotective effects before and after cerebral ischemia in female rats; Brain Res.?1474 82 Blackburn et al. (2018),?Fulvestrant for the treatment of advanced breast cancer; Expert Rev. Anticancer Ther. 18 619
Properties of Fulvestrant
| Melting point: | 104-106°C |
| Boiling point: | 674.8±55.0 °C(Predicted) |
| Density | 1.201±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | DMSO: >5mg/mL |
| form | powder |
| pka | 10.27±0.70(Predicted) |
| color | White |
| Stability: | Stable for 2 years as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 3 months. |
| CAS DataBase Reference | 129453-61-8(CAS DataBase Reference) |
Safety information for Fulvestrant
| GHS Hazard Statements |
H413:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P273:Avoid release to the environment. P501:Dispose of contents/container to..… |
Computed Descriptors for Fulvestrant
| InChIKey | VWUXBMIQPBEWFH-WCCTWKNTSA-N |
| SMILES | [C@@]12([H])[C@H](CCCCCCCCCS(=O)CCCC(F)(F)C(F)(F)F)CC3C=C(O)C=CC=3[C@@]1([H])CC[C@]1(C)[C@H](CC[C@@]21[H])O |&1:0,2,32,36,38,41,r| |
Fulvestrant manufacturer
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
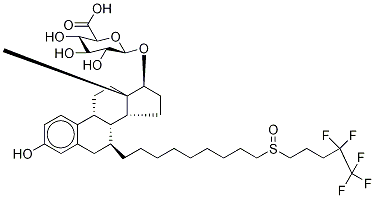
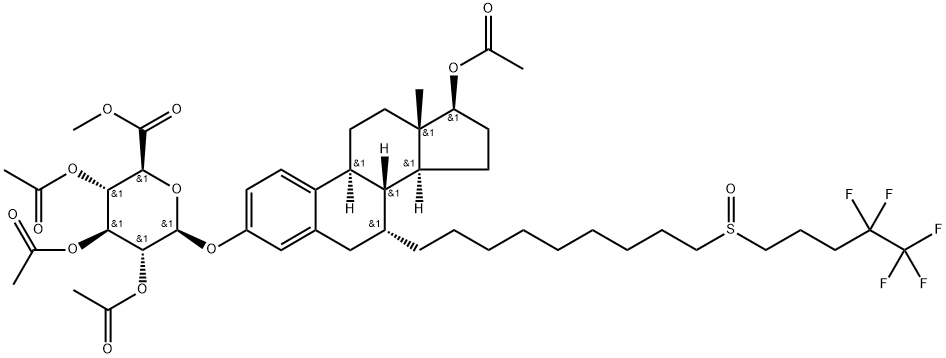
![(7a,17b)-7-7-[9-[(4,4,5,5,5-Pentafluoropentyl)sulfinyl]nonyl]-estra-1,3,5(10)-triene-3,17-diol 17-acetate](https://img.chemicalbook.in/CAS/20180808/GIF/261506-24-5.gif)

You may like
-
 129453-61-8 Fulvestrant 98%View Details
129453-61-8 Fulvestrant 98%View Details
129453-61-8 -
 Fulvestrant 99%View Details
Fulvestrant 99%View Details -
 Fulvestrant 98%View Details
Fulvestrant 98%View Details
129453-61-8 -
 Fulvestrant 129453-61-8 98%View Details
Fulvestrant 129453-61-8 98%View Details
129453-61-8 -
 Fulvestrant CAS 129453-61-8View Details
Fulvestrant CAS 129453-61-8View Details
129453-61-8 -
 Fulvestrant 95.00% CAS 129453-61-8View Details
Fulvestrant 95.00% CAS 129453-61-8View Details
129453-61-8 -
 Fulvestrant ApiView Details
Fulvestrant ApiView Details
129453-61-8 -
 Fulvestrant 5 ml Injection, SamarthView Details
Fulvestrant 5 ml Injection, SamarthView Details
129453-61-8
