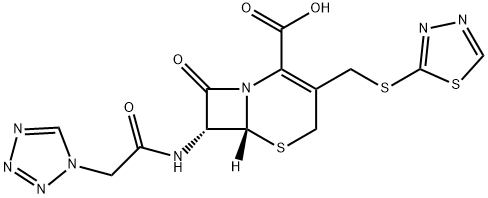
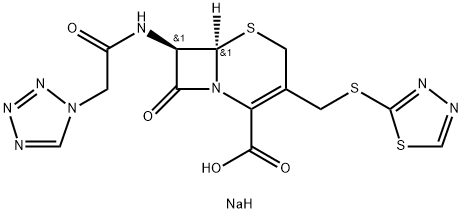
Ceftezole
Synonym(s):CZT
- CAS NO.:26973-24-0
- Empirical Formula: C13H12N8O4S3
- Molecular Weight: 440.48
- MDL number: MFCD00274176
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-21 10:59:17
What is Ceftezole?
Originator
Ceftezole,Arocor Holdings Inc.
The Uses of Ceftezole
Ceftezole is a β-lactam, cephem antibiotic. It also exhibits potent α-glucosidase inhibitory activity and antidiabetic effect in streptozotocin-induced mouse.
The Uses of Ceftezole
A direct bandgap semiconductor, it is used in a variety of applications, including radiation detectors, photorefractive gratings, electro-optic modulators, solar cells, and terahertz generation and detection. The band gap varies from approximately 1.4 to 2.2 eV, depending on composition.
Definition
ChEBI: Ceftezole is a first-generation cephalosporin antibiotic having (1,3,4-thiadiazol-2-ylsulfanyl)methyl and [2-(1H-tetrazol-1-yl)acetamido side groups located at positions 3 and 7 respectively. It is a cephalosporin and a member of thiadiazoles.
What are the applications of Application
Ceftezole is a product for proteomics research
Manufacturing Process
To a solution of 1 equivalent (eq.) of 1H-tetrazole-1-acetic acid and 1 eq. of
triethylamine in 20 ml of tetrahydrofuran cooled to -20°C was added 1 eq. of
pivaloyl chloride. After thirty-minute stirring of the mixture 20 ml of a
chloroform solution containing 1 eq. of and 1 eq. of triethylamine was poured
into the solution cooled at -10°C during a period of 30 minutes. The resulting
mixed solution was stirred for 30 minutes at the same temperature, for 1 hour
in an ice-water mixture and for 3 hours at room temperature. Removal of a
solvent from the reaction mixture afforded an oily residue, which was
dissolved into 15 ml of 10% sodium bicarbonate aqueous solution. The
resulting aqueous layer was adjusted to pH 1.0-2.0 with 10% hydrochloric
acid, washed with ether and extracted with ethyl acetate. The extract was
washed with water, dried over sodium sulfate and concentrated under reduced
pressure leaving a residue which was triturated with ethyl acetate to obtain 3-
acetoxymethyl-8-oxo-7-(2-tetrazol-1-acetylamino)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid.
A solution of 1 eq. of sodium salt of 3-acetoxymethyl-8-oxo-7-(2-tetrazol-1-
acetylamino)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid (it may be
prepared with equivalent of sodium bicarbonate and above named acid) and 1
eq. of [1.3.4]-thiadiazole-2-thiol in 20 ml of phosphate buffer (pH 6.4) was
stirred for 5.5 hours at 60°C. The reaction mixture was adjusted to pH 2.0
with 5% hydrochloric acid and treated with ethyl acetate to form a title
compound - ceftezole.
Therapeutic Function
Antibiotic
General Description
Cadmium zinc telluride (Cd1-xZnxTe or CZT) can detect nuclear radiation hence, CZT with Zn concentrations between 0.1-0.2 may be used in the making of radiation detectors of X-ray and gamma-ray spectrometers.
Properties of Ceftezole
| Melting point: | 155°C (dec.) |
| Density | 2.09 |
| storage temp. | Store at -20°C |
| solubility | DMSO: 88 mg/mL (199.78 mM);; |
| pka | 2.60±0.50(Predicted) |
| λmax | 273nm(Phosphate buffer sol.)(lit.) |
| Merck | 14,1948 |
| CAS DataBase Reference | 26973-24-0(CAS DataBase Reference) |
Safety information for Ceftezole
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07  Environment GHS09 |
| GHS Hazard Statements |
H410:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. |
Computed Descriptors for Ceftezole
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 3,4-Dibenzyloxybenzaldehyde 4-Hydrazinobenzoic acid 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 4-HYDROXY BENZYL ALCOHOL 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) 3-NITRO-2-METHYL ANILINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride tert-butyl 4- (ureidomethyl)benzylcarbamate diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
You may like
-
 Cadmium zinc telluride CASView Details
Cadmium zinc telluride CASView Details -
 55441-95-7 99%View Details
55441-95-7 99%View Details
55441-95-7 -
 N-Vinylformamide 99%View Details
N-Vinylformamide 99%View Details
13162-05-5 -
 Chloro Uracil 1820-81-1 99%View Details
Chloro Uracil 1820-81-1 99%View Details
1820-81-1 -
 207557-35-5 99%View Details
207557-35-5 99%View Details
207557-35-5 -
 2-ethyl-6-methyl-3-hydroxypyridine succinate 99%View Details
2-ethyl-6-methyl-3-hydroxypyridine succinate 99%View Details
127464-43-1 -
 2-ETHYLPYRIDINE 100-71-0 99%View Details
2-ETHYLPYRIDINE 100-71-0 99%View Details
100-71-0 -
 181228-33-1 (S)-Methyl 3-amino-2-((tert-butoxycarbonyl)amino)propanote Hydrochloride (DAP-OMe. HCl) 99%View Details
181228-33-1 (S)-Methyl 3-amino-2-((tert-butoxycarbonyl)amino)propanote Hydrochloride (DAP-OMe. HCl) 99%View Details
181228-33-1