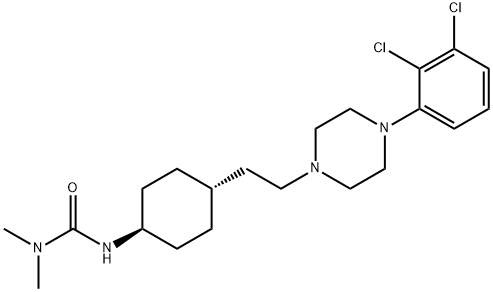
Cariprazine hydrochloride
- CAS NO.:1083076-69-0
- Empirical Formula: C21H33Cl3N4O
- Molecular Weight: 463.87
- MDL number: MFCD26142670
- EINECS: 200-077-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-12 18:21:52
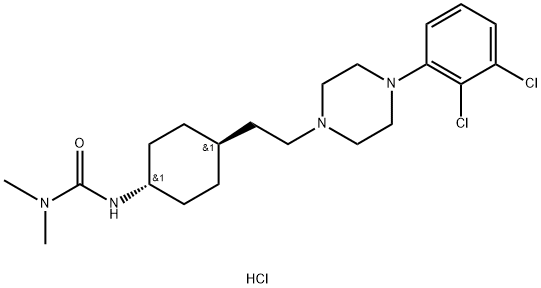
What is Cariprazine hydrochloride?
Description
Cariprazine hydrochloride (IX) is an oral, brain-penetrant, atypical antipsychotic developed by the Hungarian pharmaceutical firm Gedeon Richter. It was approved by the FDA in September 2015 for treatment of schizophrenia and for the acute treatment of manic or mixed episodes of bipolar I disorder. While the precise mechanism of action of cariprazine is unknown, its antipsychotic and procognitive effects may be mediated through partial agonism at dopamine D2/D3 and serotonin 5-HT1A receptors as well as antagonism at serotonin 5-HT2A receptors. Unlike many antipsychotics, cariprazine displays particular selectivity for the D3 receptor (D3, Ki = 0.085 nM; D2L, Ki = 0.49 nM; D2S, Ki = 0.69 nM). Cariprazine is extensively metabolized by CYP3A4 and, to a lesser extent, CYP2D6; desmethyl and didesmethyl cariprazine, the primary metabolites, are pharmacologically equipotent to the parent drug. In clinical trials, cariprazine demonstrated improvement compared to placebo as measured by Young Mania Rating Scale (YMRS) total scores in patients with bipolar mania and by Positive and Negative Syndrome Scale (PANSS) total scores in patients with schizophrenia. Forest Laboratories (now Allergan) has exclusive rights to cariprazine in the U.S. and Canada, while Mitsubishi Pharma Corporation has exclusive rights to the sale of the drug in Japan and Asia.
The Uses of Cariprazine hydrochloride
Cariprazine is used to treat schizophrenia (a mental illness that causes disturbed or unusual thinking, loss of interest in life, and strong or inappropriate emotions). Cariprazine is also used to treat episodes of depression in people with bipolar I disorder (manic depressive disorder; a disease that causes episodes of mania, episodes of depression and other abnormal moods). It is also used as a short term treatment for episodes of mania or mixed episodes (symptoms of mania and depression that happen together) in people with bipolar I disorder. Cariprazine is in a class of medications called atypical antipsychotics. It works by changing the activity of certain natural substances in the brain.
Definition
ChEBI: Cariprazine hydrochloride is a hydrochloride obtained by combining cariprazine with one molar equivalent of hydrochloric acid. Used for treatment of schizophrenia and bipolar disorder. It has a role as a second generation antipsychotic, a dopamine agonist and a serotonergic antagonist. It contains a cariprazine(1+).
Side Effects
Common side effects of Cariprazine hydrochloride include: extreme tiredness, restlessness and anxiety, difficulty sleeping, dizziness, feeling unsteady or difficulty in maintaining balance; increased appetite or weight gain; constipation, dyspepsia, nausea, increased salivation or drooling. More serious side effects may include: uncontrollable unusual movements of the body or face; slow or shuffling walking; loss of mobility, fever, sweating, confusion, shortness of breath, fast or irregular heartbeat; and severe muscle stiffness; muscle weakness or soreness; blank facial expression; difficulty swallowing or breathing; tightness in the throat; tongue spitting, rash, itching, measles; face, throat, tongue, lips, or eyes are swollen; dark or cola-coloured urine; swelling of the legs and feet; and decreased urination.
Synthesis
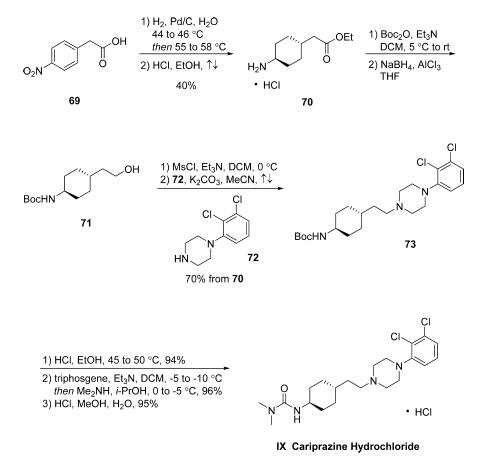
While the synthesis of cariprazine hydrochloride has been reported in a number of patents as well as its discovery synthesis in the publicly disclosed literature, the process route has not yet been disseminated. Starting with the reduction of commercial 2-(4- nitrophenyl)acetic acid (69) via hydrogenation in water in the presence of Pd/C, this reaction proceeds a one-pot, stepwise reduction of the nitro group. A separate reduction event converting the phenyl ring to the corresponding cyclohexane provides 4-aminocyclohexylacetic acid with 60-70% selectivity for the desired trans isomer. Following filtration and distillation, the crude aqueous solution was treated with HCl in refluxing ethanol to generate the corresponding ethyl ester 70. Crystallization from acetonitrile gave the HCl salt in high purity and 40% yield over two steps (a reaction sequence that was reported on 200 kg scale). Amine 70 was transformed into intermediate 73 via Boc protection followed by ester reduction to the primary alcohol 71, which was obtained as a solution in toluene following extraction. Next, mesylation of the alcohol followed by alkylation with commercially available piperazine 72 provided piperazinyl cyclohexane 73 in 70% over the four-step sequence. The carbamate protecting group within 73 was removed via acidic ethanolysis, and the resulting product was treated with triphosgene and dimethylamine to generate cariprazine as the freebase. Salt formation by means of methanolic HCl ultimately furnished cariprazine hydrochloride IX in 85% yield from 73.
Properties of Cariprazine hydrochloride
| storage temp. | Store at -20°C |
| solubility | DMSO : 6.67 mg/mL (14.38 mM; Need ultrasonic) |
| form | Powder |
| color | White to off-white |
| InChI | InChI=1/C21H32Cl2N4O.ClH/c1-25(2)21(28)24-17-8-6-16(7-9-17)10-11-26-12-14-27(15-13-26)19-5-3-4-18(22)20(19)23;/h3-5,16-17H,6-15H2,1-2H3,(H,24,28);1H/t16-,17-; |
Safety information for Cariprazine hydrochloride
Computed Descriptors for Cariprazine hydrochloride
| InChIKey | GPPJWWMREQHLQT-BHQIMSFRNA-N |
| SMILES | ClC1C(=CC=CC=1N1CCN(CC[C@@H]2CC[C@@H](NC(=O)N(C)C)CC2)CC1)Cl.Cl |&1:13,16,r| |
Cariprazine hydrochloride manufacturer
Aquigen Bio science Pvt Ltd
Archerchem Healthcare Pvt., Ltd. (part of Archerchem Group)
New Products
Indole Methyl Resin tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Boc-His(Boc)-OH 2-CTC Resin 4-Chloro-7-tosy1-7Hpyrrolo[2,3-d]pyrimidine 5,7-Dibromo-1H-indole 2,5-dichloro-N-hydroxy-4,6-dimethylpyridine-3-carboximidamide 2,2-Dimethoxy-7-azaspiro[3.5]nonane hydrochloride 4-chloromethyl-5-methyl-1,3-dioxol-2-one (DMDO-Cl) R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDIIMIDAZOLE 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID 4-((2-hydroxyethyl)thio)benzoic acid 1-(TERT-BUTOXYCARBONYL)-2-PYRROLIDINONE Methyl 6-methylnicotinate 3-Pyridineacrylic acid tert-Butyl carbazate TETRAHYDRO-2H-PYRAN-3-OL 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile 2,4-dihydroxybenzaldehyde 1,3-Diethyl-1,3-Diphenylurea Methyl 2-methylquinoline-6-carboxylateRelated products of tetrahydrofuran
You may like
-
 CARIPRAZINE HYDROCHLORIDE 98%View Details
CARIPRAZINE HYDROCHLORIDE 98%View Details -
 1083076-69-0 98%View Details
1083076-69-0 98%View Details
1083076-69-0 -
 1083076-69-0 98%View Details
1083076-69-0 98%View Details
1083076-69-0 -
 1083076-69-0 Cariprazine Hydrochloride 97 %View Details
1083076-69-0 Cariprazine Hydrochloride 97 %View Details
1083076-69-0 -
 Pyridine 99.5% HPLC /UV SpectroscopyView Details
Pyridine 99.5% HPLC /UV SpectroscopyView Details
110-86-1 -
 Dibutyl PhthalateView Details
Dibutyl PhthalateView Details
84-74-2 -
 Imidazole Spot supply, competitive priceView Details
Imidazole Spot supply, competitive priceView Details
288-32-4 -
 Thiourea 99% ARView Details
Thiourea 99% ARView Details
62-56-6