Boxidine
- CAS NO.:10355-14-3
- Empirical Formula: C19H20F3NO
- Molecular Weight: 335.36
- MDL number: MFCD00865721
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-10-28 23:16:16
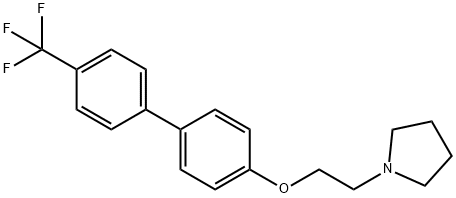
What is Boxidine?
Originator
Boxidine,Onbio Inc.
The Uses of Boxidine
Antihyperlipoproteinemic.
Definition
ChEBI: Boxidine is a member of biphenyls.
Manufacturing Process
The title compound may be prepared 2 ways:
1). 1-(2-[4'-(Trifluoromethyl)-4-biphenylyloxy]ethyl)pyrrolidine:
A suspension consisting of 89.8 g (0.33 mole) of p-iodobenzotrifluoride, 132.5
g (0.65 mole) of p-iodoanisole, and 322.7 g of Cu powder in DMF (175 ml)
was heated (225-230°C) with stirring in a resin pot for about 5 days. After
cooling, the solid reaction mass was pulverized and continuously extracted
(heptane) for 2 days. Evaporation of the solvent left a dark brown residue (ca.
50 g) which was dissolved (heptane, 200 ml), decolorized (charcoal), and
concentrated to 100 ml. On standing ca. 20 g of impure 4,4'-
dimethoxybiphenyl were deposited as colorless crystals. Fractional
crystallization was continued until the crops of crystalline material were free of
impurities by TLC (80:20 heptane-ethyl acetate). Pure 4-methoxy-4'-
trifluoromethylbiphenyl was isolated as colorless granules, 21.6 g (26%), MP:
124-126°C. A solution consisting of 21.6 g (0.09 moles) of 4-methoxy-4'-
trifluoromethylbiphenyl dissolved in glacial acetic acid and HBr (48%) was
refluxed for approximately 24 hr. After cooling the acetic reaction mixture was
poured into H2O (1.5 L) and the solid which separated was collected and air
dried. 18.0 g (83%) of the crude product was isolated and taken up in Et2O
(100 ml), decolorized (charcoal), filtered, and concentrated to one-third of the
original volume. The material which separated from the Et2O solution (m.p.
147-148°C) was pure enough for the next synthetic step (structure verified by
NMR). 1.5.6 g (0.06 mole) of the sodium derivative of 4-hydroxy-4'-
trifluoromethylbiphenyl (prepared from 4-hydroxy-4'-trifluoromethylbiphenyl
and sodium hydride) allowed to react with 8.0 g (0.06 mole) of 2-
pyrrolidinylethyl chloride in refluxing DMF (100 ml) for 18 hr. The resulting
suspension was cooled, filtered, and the clear filtrate was concentrated to
semisolid residue. Two 100 ml portions of water were used to triturate the
crude product which was then dissolved in benzene; the solution was
decolorized (char coal) and dried (Na2SO4) and the benzene was removed.
Several fractional crystallizations from acetone afforded 9.8 g (49%) of pure
1-1 2- [4'-(trifluoromethyl)-4-biphenylyloxy]ethyl)pyrrolidine MP: 109-110°C.
2). p-Bromobenzotrifluoride (1.37 g, 0.7 mole) and ca. 1.0 g of MeI dissolved
in dry Et2O (200 ml) was added to 19 g (0.8 g-atom) of 11 g suspended in
Et2O (20 ml) under the usual conditions. Addition of the aromatic halide was
regulated to maintain a gentle reflux and refluxing was continued an additional 1 hr after addition was complete. 4-Methoxycyclohexanone (64 g,
0.5 mole) dissolved in 75 ml of dry Et2O was added to the freshly formed
Grignard reagent with vigorous stirring and, after addition of the ketone was
complete, the reaction mixture was refluxed with stirring for approximately 1
hr. Decomposition of the Grignard reagent-ketone addition product was
achieved by adding excess cold, aqueous ammonia chloride (53 g in 1 L of
H2O), and the crude product was removed using two 100-ml portions of Et2O.
The combined extracts were decolorized (charcoal), filtered, and dried
(Na2SO4). Removal of the Et2O left a brown, oily residue which was distilled in
vacuum affording 51.3 g (38%) of 1-(p-trifluoromethylphenyl)-4-
methoxycyclohexanol, b.p. 121-122°C (0.4-0.5 mm), m.p. 53-54°C.
The 4-methoxycyclohexanol derivative (27 g, 0.1 mole), purified as described
above, was added to a vigorously stirred concentrated H2SO2-glacial acetic
acid (10:40 ml) solution. When a clear solution resulted (ca. 2 min), the
reaction mixture was poured all at once into a previously cooled (5-10%
mixture of H2O (300 ml) covered with Et2O (300 ml)). The Et2O layer was
separated, dried (Na2SO4), and concentrated to a brown, oily residue.
Fractionation of the crude oil yielded 18.3 g (717) of 1-(ptrifluoromethylphenyl)-
4-methoxycyclohexene, b.p. 104-103°C (0.3-0.4 mm).
Dehydrogenation of the purified 4-methoxycyclohexene derivative, obtained as
described above, was accomplished using a modification of the method
described by Anisworth (J.A.C.S., 76, 4446 (1954)). A suspension consisting
of 1-(p-trifluoromethylphenyl)-4-methoxycyclohexene (500 g, 1.95 moles),
166 g of Pd/C and nitrobenzene was refluxed for 22 hr. Aliquots of the
reaction mixture taken periodically and analyzed by TLC (heptane-ethyl
acetate (4:1) solvent system) indicated that aromatization was complete after
this period of time, Removal of the nitrobenzene under reduced pressure left
442 g (89.9%) of the crude biphenyl derivative. Two recrystallizations
(petroleum ether) produced material identical with that obtained from the 1
procedure described above.
Therapeutic Function
Antihyperlipidemic
Properties of Boxidine
| Melting point: | 108-110 °C(Solv: methanol (67-56-1); water (7732-18-5)) |
| Boiling point: | 415.4±45.0 °C(Predicted) |
| Density | 1.178±0.06 g/cm3(Predicted) |
| pka | 9.56±0.20(Predicted) |
Safety information for Boxidine
Computed Descriptors for Boxidine
New Products
4-Fluorophenylacetic acid 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate (6-METHYL-[1,3]DITHIOLO[4,5-b]QUINOXALIN-2-ONE INDAZOLE-3-CARBOXYLIC ACID 4-IODO BENZOIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
![4'-(TRIFLUOROMETHYL)[1,1'-BIPHENYL]-4-OL](https://img.chemicalbook.in/CAS/GIF/10355-13-2.gif)
![[2-(BIPHENYL-4-YLOXY)-ETHYL]-DIMETHYL-AMINE](https://img.chemicalbook.in/CAS/GIF/52402-78-5.gif)

You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1