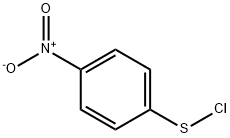
2,4-DINITROBENZENESULFENYL CHLORIDE
Synonym(s):2,4-Dinitrophenylsulfenyl chloride;Kharasch reagent
- CAS NO.:528-76-7
- Empirical Formula: C6H3ClN2O4S
- Molecular Weight: 234.62
- MDL number: MFCD00007129
- EINECS: 208-441-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:15:30
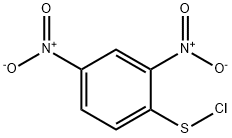
What is 2,4-DINITROBENZENESULFENYL CHLORIDE?
The Uses of 2,4-DINITROBENZENESULFENYL CHLORIDE
2,4-Dinitrobenzenesulfenyl chloride was used in preparation of chlorine and bromine biphenyls.
Preparation
CAUTION: This reaction should be carried out in a hood behind a safety shield. The product decomposes explosively if heated above 90-100°C .
To a 500-ml, three-necked flask equipped with a mechanical stirrer, distillation column, and distillation head is added 100 gm (0.25 mole) of dry 2,4-dinitrophenyl disulfide and 250 ml of ethylene chloride. The contents are heated with an oil bath at 125°C until 30-40 ml of the solvent is collected by distillation. The contents are cooled and the distillation head replaced by a gas inlet and exit tube connected to an alkali trap. Then 0.15 gm (0.002 mole) of Merck "Iron by Hydrogen" powder is added as a catalyst [51a-c], and dry chlorine gas is slowly bubbled into the reaction. In a few minutes, the reaction warms up, and the flask is surrounded with a 20°C water bath. The chlorine gas is bubbled in at such a rate that 18 gm (0.25 mole) is added over a 1-1½-hr period. After this time, the suspended disulfide should gradually disappear, and a clear, amber-red solution results. The flow of chlorine gas is stopped, the solution stirred for 1 hr, filtered through diatomaceous earth, the filtrate aspirated to remove excess chlorine, and then 400 ml of anhydrous ether is added. Within ½ hr, most of the product crystallizes by cooling the flask in an ice-salt-water bath for 3-4 hr. The product is filtered, washed with 100 ml of cold ether, and then dried to afford 70 gm (60%) of brilliant yellow needles, m.p. 95-95.5°C.
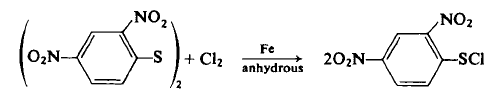
Synthesis Reference(s)
Journal of the American Chemical Society, 72, p. 1796, 1950 DOI: 10.1021/ja01160a108
General Description
2,4-Dinitrobenzenesulfenyl chloride undergoes addition reactions with transannularly substituted cycloalkenes and norbornenes. 2,4-Dinitrobenzenesulfenyl chloride can convert allylic alcohols to dienes by 1,4-elimination.
Purification Methods
Crystallise the sulfenyl chloride from CCl4. [Beilstein 6 II 316.]
Properties of 2,4-DINITROBENZENESULFENYL CHLORIDE
| Melting point: | 94-97 °C(lit.) |
| Boiling point: | 430.1±35.0 °C(Predicted) |
| Density | 1.700 (estimate) |
| refractive index | 1.6000 (estimate) |
| storage temp. | 2-8°C |
| solubility | benzene: soluble |
| form | solid |
| color | Light orange to Yellow to Green |
| Merck | 14,3274 |
| BRN | 405600 |
| CAS DataBase Reference | 528-76-7(CAS DataBase Reference) |
| EPA Substance Registry System | Benzenesulfenyl chloride, 2,4-dinitro- (528-76-7) |
Safety information for 2,4-DINITROBENZENESULFENYL CHLORIDE
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H228:Flammable solids H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P240:Ground/bond container and receiving equipment. P241:Use explosion-proof electrical/ventilating/lighting/…/equipment. P260:Do not breathe dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P310:Immediately call a POISON CENTER or doctor/physician. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P370+P378:In case of fire: Use … for extinction. P405:Store locked up. P501:Dispose of contents/container to..… |
Computed Descriptors for 2,4-DINITROBENZENESULFENYL CHLORIDE
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 4-Hydrazinobenzoic acid 3,4-Dibenzyloxybenzaldehyde Electrolytic Iron Powder 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 4-HYDROXY BENZYL ALCOHOL 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) S-2-CHLORO PROPIONIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 3-(Hydroxymethyl)benzoate N-Boc-2-chloroethylamine 1-Bromo-2-methoxy-3-nitrobenzene N-Methyl-3-cyclopenten-1-amine 2-Bromo-3-hydroxybenzaldehyde 1H-indazole-5-carboxamideRelated products of tetrahydrofuran

You may like
-
 2,4-Dinitrophenylsulfenyl Chloride CAS 528-76-7View Details
2,4-Dinitrophenylsulfenyl Chloride CAS 528-76-7View Details
528-76-7 -
 7441-43-2 98%View Details
7441-43-2 98%View Details
7441-43-2 -
 1260741-78-3 6-Bromo-3-iodo-1-methyl-1H-indazole 98%View Details
1260741-78-3 6-Bromo-3-iodo-1-methyl-1H-indazole 98%View Details
1260741-78-3 -
 4-bromo-3,5-dimethylbenzenesulfonyl chloride 1581266-79-6 98%View Details
4-bromo-3,5-dimethylbenzenesulfonyl chloride 1581266-79-6 98%View Details
1581266-79-6 -
 2490430-37-8 98%View Details
2490430-37-8 98%View Details
2490430-37-8 -
 N-(5-Amino-2-methylphenyl)acetamide 5434-30-0 98%View Details
N-(5-Amino-2-methylphenyl)acetamide 5434-30-0 98%View Details
5434-30-0 -
 124371-59-1 98%View Details
124371-59-1 98%View Details
124371-59-1 -
 53857-52-2 98%View Details
53857-52-2 98%View Details
53857-52-2