1,10-Phenanthroline hydrate
Synonym(s):o-Phenanthroline - CAS 5144-89-8 - Calbiochem;o-Phenanthroline monohydrate;1,10-Phenanthroline monohydrate;o-Phenanthroline monohydrate
- CAS NO.:5144-89-8
- Empirical Formula: C12H8N2.H2O
- Molecular Weight: 198.22
- MDL number: MFCD00149973
- EINECS: 627-114-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-23 21:30:31
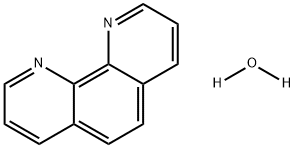
What is 1,10-Phenanthroline hydrate?
Chemical properties
white crystals or powder
The Uses of 1,10-Phenanthroline hydrate
1,10-Phenanthroline monohydrate is useful to study the DNA-protein interactions due to its nuclease activity. It is used as a redox indicator and reagent for spectrophotometric assay of silver. It acts as a chelating ligand for the determination of Fe, Pd and V. It also acts as a matrix metalloproteinase inhibitor. It is a photometric reagent for iron, which forms a red chelate with ferrous (iron II) ion.
The Uses of 1,10-Phenanthroline hydrate
When complexed with copper, it possesses nuclease activity that has been used to study DNA-protein interactions.
1,10-Phenanthroline monohydrate is an aromatic nitrogen heterocycle that may be used as a chelating agent in the preparation of metal-chelate complexes.
What are the applications of Application
o-Phenanthroline monohydrate is a chelating agent for Fe2+
General Description
White crystalline powder or solid. Slight odor.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
1,10-Phenanthroline hydrate is incompatible with strong oxidizers. 1,10-Phenanthroline hydrate is also incompatible with strong acids. 1,10-Phenanthroline hydrate forms complex compounds with ferrous ions. .
Fire Hazard
Flash point data for 1,10-Phenanthroline hydrate are not available. 1,10-Phenanthroline hydrate is probably combustible.
Biochem/physiol Actions
Metalloprotease inhibitor; chelates iron, zinc and other divalent metals. Effective concentration 1-10 mM.
Purification Methods
Crystallise its picrate (m 191o) from EtOH; then the free base is liberated with aqueous alkali, dried at 78o/8mm over P2O5 and crystallised from pet ether (b 80-100o). [Cumper et al. J Chem Soc 1188 1962.] It can be purified by zone melting. It has also been crystallised from hexane, *benzene/pet ether (b 40-60o) or sodium-dried *benzene, dried and stored over H2SO4. The monohydrate is obtained by crystallisation from aqueous EtOH or ethyl acetate. It has been crystallised from H2O (300 parts) to give the monohydrate m 102-103o which sublimes at 10-3mm [Fielding & LeFevre J Chem Soc 1811 1951.] The anhydrous compound has m 118o (after drying at high vacuum at 80o) and is also obtained by recrystallisation from pet ether or *C6H6 (70 parts) and drying at 78o/8mm. [UV: Badger et al. J Chem Soc 3199 1951.] IthasapKainH2Oof 4.857 (25o) or 5.02 (20o) and 4.27 in 50% aqueous EtOH (20o). [Albert et al. J Chem Soc 2240 1948]. [Beilstein 23 H 227, 23 II 235, 23/8 V 419.]
Properties of 1,10-Phenanthroline hydrate
| Melting point: | 100-104 °C(lit.) |
| Density | 1.10 |
| vapor density | 6.2 (vs air) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | methanol: soluble200MM (Stock solution is stable for months at –20° C.) |
| form | Needle-Like Crystals |
| color | Off-white, may become cream-colored during storage |
| Water Solubility | <0.01 g/100 mL at 21 ºC |
| Merck | 14,7214 |
| BRN | 4008096 |
| Stability: | Stable. Incompatible with iron, other heavy metals, strong oxidizing agents, strong acids. |
| CAS DataBase Reference | 5144-89-8(CAS DataBase Reference) |
| EPA Substance Registry System | 1,10-Phenanthroline, monohydrate (5144-89-8) |
Safety information for 1,10-Phenanthroline hydrate
| Signal word | Danger |
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06  Environment GHS09 |
| GHS Hazard Statements |
H301:Acute toxicity,oral H410:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P273:Avoid release to the environment. |
Computed Descriptors for 1,10-Phenanthroline hydrate
1,10-Phenanthroline hydrate manufacturer
Suvchem
New Products
Indole Methyl Resin tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Boc-His(Boc)-OH 2-CTC Resin 4-Chloro-7-tosy1-7Hpyrrolo[2,3-d]pyrimidine 5,7-Dibromo-1H-indole 2,5-dichloro-N-hydroxy-4,6-dimethylpyridine-3-carboximidamide 2,2-Dimethoxy-7-azaspiro[3.5]nonane hydrochloride 4-chloromethyl-5-methyl-1,3-dioxol-2-one (DMDO-Cl) R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDIIMIDAZOLE 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID 4-((2-hydroxyethyl)thio)benzoic acid 1-(TERT-BUTOXYCARBONYL)-2-PYRROLIDINONE Methyl 6-methylnicotinate 3-Pyridineacrylic acid tert-Butyl carbazate TETRAHYDRO-2H-PYRAN-3-OL 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile 2,4-dihydroxybenzaldehyde 1,3-Diethyl-1,3-Diphenylurea Methyl 2-methylquinoline-6-carboxylateRelated products of tetrahydrofuran


You may like
-
 1,10-Phenanthroline Monohydrate extrapure AR CAS 5144-89-8View Details
1,10-Phenanthroline Monohydrate extrapure AR CAS 5144-89-8View Details
5144-89-8 -
 1,10-Phenanthroline CAS 5144-89-8View Details
1,10-Phenanthroline CAS 5144-89-8View Details
5144-89-8 -
![1,10-Phenanthroline Monohydrate [for Biochemical Research] CAS 5144-89-8](https://img.chemicalbook.in//Content/image/CP5.jpg) 1,10-Phenanthroline Monohydrate [for Biochemical Research] CAS 5144-89-8View Details
1,10-Phenanthroline Monohydrate [for Biochemical Research] CAS 5144-89-8View Details
5144-89-8 -
 1,10-Phenanthroline, GR 99%+ CAS 5144-89-8View Details
1,10-Phenanthroline, GR 99%+ CAS 5144-89-8View Details
5144-89-8 -
 1,10 Phenanthroline Monohydrate >99.5% (CAS : 5144-89-8) RS.15000=00 PER 1KG 18% GSTView Details
1,10 Phenanthroline Monohydrate >99.5% (CAS : 5144-89-8) RS.15000=00 PER 1KG 18% GSTView Details
5144-89-8 -
 1,10-Phenanthroline MonohydrateView Details
1,10-Phenanthroline MonohydrateView Details
5144-89-8 -
 1,10 Phenanthroline MonohydrateView Details
1,10 Phenanthroline MonohydrateView Details
5144-89-8 -
 1,10 Phenanthroline MonohydrateView Details
1,10 Phenanthroline MonohydrateView Details
5144-89-8
