1-beta-D-Arabinofuranosylcytosine hydrochloride
Synonym(s):Cytarabine;Arabinosylcytosine;Ara-C;Cytarabine hydrochloride;Cytosine arabinoside
- CAS NO.:69-74-9
- Empirical Formula: C9H14ClN3O5
- Molecular Weight: 279.68
- MDL number: MFCD00012839
- EINECS: 200-713-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:07:02
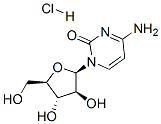
What is 1-beta-D-Arabinofuranosylcytosine hydrochloride?
Description
Cytosine beta-D-arabinofuranoside Hydrochloride is the salt form of Cytarabine, which is a selective inhibitor of DNA synthesis and is used as an antineoplastic and antiviral. Cytosine beta-D-arabinofuranoside Hydrochloride is used in chemotherapy for the treatment of white blood cells cancers such as acute myeloid leukemia (AML) and non-Hodgkin lymphoma.
Chemical properties
Crystalline
Originator
Cytosar,Upjohn,US,1969
The Uses of 1-beta-D-Arabinofuranosylcytosine hydrochloride
Antiviral.
The Uses of 1-beta-D-Arabinofuranosylcytosine hydrochloride
Interferes with the synthesis of DNA
What are the applications of Application
Cytosine β-D-arabinofuranoside hydrochloride is a DNA synthesis inhibitor and anti-leukemic agent
Manufacturing Process
(A) Preparation of 1- (2,3,5-Tri-O-Acetyl-β-D-Arabinofuranosyl)-4-Thiouracil: A
mixture of 1.85 g (5.0 mmol) of 1-(2,3,5-tri-O-acetyl-β-arabinofuranosyl)
uracil, 1.23 g (5.55 mmol) of phosphorus pentasulfide, and 30 ml of pyridine
was heated under gentle reflux for 2.5 hours with exclusion of moisture. The
reaction mixture was cooled, and the supernatant solution was transferred by
means of a pipette into a mixture of crushed ice and water. The reaction flask
was washed twice with pyridine, and these washings were added to the icewater mixture. This mixture was kept at about 25°C until the ice had melted,
and was then stored at 0°C for one hour. A pale yellow precipitate that formed
was collected on a filter, washed with ice-water, and dried in air.
This material was triturated with chloroform, and the chloroform mixture was
filtered. A small amount of undissolved material collected on the filter and it was washed with chloroform. The chloroform solution (filtrate plus washings)
was washed three times with ice-water, twice with ice-cold 3 N sulfuric acid,
twice with ice-cold saturated aqueous sodium bicarbonate solution, twice with
ice-water, and then dried over anhydrous sodium sulfate. The chloroform was
removed under reduced pressure at a bath temperature of about 40°C,
leaving a yellow, somewhat gummy residue. This yellow residue was dissolved
in absolute methanol which was then evaporated at reduced pressure at about
40°C, and the residue was then held for 2 hours at 0.5 to 2.0 mm pressure
and a bath temperature of about 50°C. There was thus obtained 1.69 g of 1-
(2,3,5-tri-O-acetyl-β-D-arabinofuranosyl)-4-thiouracil.
(B) Preparation of 1-β-D-Arabinofuranosylcytosine: In a glass liner, a mixture
of 1.16 g (3.0 mmol) of 1-(2,3,5-tri-O-acetyl-β-D-arabinofuranosyl)-4-
thiouracil prepared in (A) and about 60 ml of absolute methanol which had
been saturated with anhydrous ammonia at 0°C was heated in a steel bomb
at 98° to 105°C for 35 hours. After cooling to about 25°C and venting the
bomb, the dark solution was filtered into a round-bottom flask. The methanol
and excess ammonia were then removed under reduced pressure at about
25°C. The residual syrup was dissolved in absolute methanol, and the
methanol was removed under reduced pressure at a bath temperature of
about 40°C. This procedure of dissolving in absolute methanol and removing
the solvent was repeated, and the residue was held under reduced pressure at
a bath temperature of 45°C for 12 hours.
The resulting semisolid was triturated thoroughly with absolute methanol, and
the resulting suspension was chilled at 0°C. A pale tan solid that separated
was collected on a filter and washed repeatedly with methanol. After washing
with anhydrous ether, there was obtained 430 mg of 1-β-Darabinofuranosylcytosine.
(C) Preparation of 1-β-D-Arabinofuranosylcytosine Hydrochloride: The absolute
methanolic filtrate obtained after triturating and filtering the 1-β-Darabinofuranosylcytosine in (B) above was warmed and stirred with
decolorizing charcoal. The mixture was filtered through a bed of filter aid, and
the filter bed was washed repeatedly with absolute methanol. The combined
filtrate and washings were pale yellow. The solution was diluted to faint
cloudiness with anhydrous ether, and an excess of anhydrous hydrogen
chloride was introduced. Crystallization began at about 25°C and further
crystallization was induced by chilling at 0°C for 14 hours. The crystalline
product was collected on a filter, washed with anhydrous ether, and dried in
air. There was thus obtained 180 mg of pale yellow 1-β-Darabinofuranosylcytosine hydrochloride melting at 186° to 189°C.
The pale yellow product was dissolved in warm, absolute methanol, and the
solution after mixing with decolorizing charcoal was filtered through a bed of
filter aid. The filter bed was washed with warm absolute methanol, and the
combined methanolic filtrate and washings were warmed and diluted with
anhydrous ether to incipient crystallization. The methanol-ether mixture was
kept at about 25°C for about 1 hour and then chilled, first at 0°C, and then at
-20°C. The resulting colorless needles were collected on a filter, washed with
anhydrous ether, and dried at 85°C, yielding 100 mg of 1-β-Darabinofuranosylcytosine hydrochloride having a melting point of 186° to
188°C.
Therapeutic Function
Cancer chemotherapy
General Description
Crystals (from aqueous ethanol) or fluffy white powder.
Air & Water Reactions
Water soluble. Hydrolysis occurs readily, especially with acid.
Reactivity Profile
1-beta-D-Arabinofuranosylcytosine hydrochloride undergoes decomposition in acidic solutions.
Fire Hazard
Flash point data for 1-beta-D-Arabinofuranosylcytosine hydrochloride are not available; however, 1-beta-D-Arabinofuranosylcytosine hydrochloride is probably combustible.
Biological Activity
cytarabine hydrochloride is an effective drug in the treatment of cancers of white blood cells [1].cytarabine hydrochloride is a dxoxycytidine (dc) analogue. cytarabine hydrochloride has been found to be phosphorylated into a triphophate form, and thus compete with dctp for incorporation into dna. cytarabine hydrochloride has reported to incorporate into dna and block dna synthesis by inhibiting the function of dna and rna polymerases. in addition, incorporated has shown a growth inhibition dose-dependent curve using acute myelogenous leukemia (aml) in a growth inhibition assay with ic50 values of 16nm,103μm and 223μm for ccrf-cem, cem/arac8c and cem/dck-cell lines, respectively. moreover, cytarabine hydrochloride has been exhibited to retrovirally transducer rat leukemic ka cells by wst-1 assay with ic50 values of 0.69μm,1.73μm and 0.037μm for ka, ka/gfp and ka/wt, respectively [1,2].
Biochem/physiol Actions
Ara-C is phosphorylated to Ara-CTP and is incorporatee into DNA. It inhibits DNA replication by forming cleavage complexes with topoisomerase I resulting in DNA fragmentation, and ultimately induces apoptosis via the PKC signaling pathway. Does not inhibit RNA synthesis. Anti-leukemia agent.
Safety Profile
Poison by intravenous route.Moderately toxic by intraperitoneal and subcutaneousroutes. Experimental reproductive effects. A human eyeirritant. Mutation data reported. When heated todecomposition it emits very toxic fumes of NOx and HCl.
References
[1] tobias sc1, borch rf.synthesis and biological evaluation of a cytarabine phosphoramidate prodrug. mol pharm. 2004 mar-apr; 1(2):112-6.
[2] veuger mj1, heemskerk mh, honders mw, willemze r, barge rm.functional role of alternatively spliced deoxycytidine kinase in sensitivity to cytarabine of acute myeloid leukemic cells. blood. 2002 feb 15; 99(4):1373-80.
Properties of 1-beta-D-Arabinofuranosylcytosine hydrochloride
| Melting point: | 197-198 °C(lit.) |
| storage temp. | 2-8°C |
| solubility | insoluble in EtOH; ≥14.2 mg/mL in DMSO; ≥64.2 mg/mL in H2O |
| form | crystalline |
| color | White to off-white |
| Stability: | Stable. Combustible. Incompatible with strong oxidizing agents. |
| CAS DataBase Reference | 69-74-9(CAS DataBase Reference) |
| EPA Substance Registry System | Cytarabine hydrochloride (69-74-9) |
Safety information for 1-beta-D-Arabinofuranosylcytosine hydrochloride
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H317:Sensitisation, Skin H319:Serious eye damage/eye irritation H341:Germ cell mutagenicity H361:Reproductive toxicity |
| Precautionary Statement Codes |
P201:Obtain special instructions before use. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P308+P313:IF exposed or concerned: Get medical advice/attention. |
Computed Descriptors for 1-beta-D-Arabinofuranosylcytosine hydrochloride
| InChIKey | KCURWTAZOZXKSJ-XKIMMOKZSA-N |
New Products
4-Fluorophenylacetic acid 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate (6-METHYL-[1,3]DITHIOLO[4,5-b]QUINOXALIN-2-ONE INDAZOLE-3-CARBOXYLIC ACID 4-IODO BENZOIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
You may like
-
 Thermo Scientific Chemicals Cytosine-beta-D-arabinofuranose hydrochloride CAS 69-74-9View Details
Thermo Scientific Chemicals Cytosine-beta-D-arabinofuranose hydrochloride CAS 69-74-9View Details
69-74-9 -
 Cytarabine HCl 97% CAS 69-74-9View Details
Cytarabine HCl 97% CAS 69-74-9View Details
69-74-9 -
 Cytosine β-D-arabinofuranoside hydrochloride CAS 69-74-9View Details
Cytosine β-D-arabinofuranoside hydrochloride CAS 69-74-9View Details
69-74-9 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1