86220-42-0
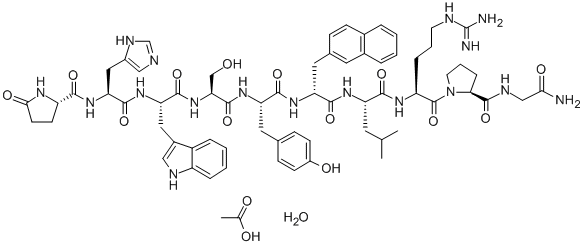
COMPUTED DESCRIPTORS
| Molecular Weight | 1400.5 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 18 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 33 |
| Exact Mass | 1399.66731982 g/mol |
| Monoisotopic Mass | 1399.66731982 g/mol |
| Topological Polar Surface Area | 513 Ų |
| Heavy Atom Count | 101 |
| Formal Charge | 0 |
| Complexity | 2760 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 9 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 3 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Nafarelin Acetate is the acetate salt form of nafarelin, a modified synthetic porcine luteinizing hormone (LH)-releasing hormone peptide analog, with gonadotropin-releasing hormone (GnRH) agonist activity. Upon nasal inhalation, nafarelin acetate binds to the GnRH receptor. This initially results in the release of the gonadotropins, follicle-stimulating hormone (FSH) and LH, from the pituitary gland; however, prolonged stimulation of the GnRH receptor desensitizes the receptor, which leads to decreased secretion of FSH and LH. In females, the inhibition of gonadotropin secretion causes hypogonadotropic hypogonadism leading to decreased production of estrogen and progesterone and anovulation. In males, the inhibition of LH secretion prevents the production and release of testosterone from Leydig cells in the testes and causes a significant decline in testosterone production that is near the levels seen following castration.