Yohimbine
- CAS NO.:146-48-5
- Empirical Formula: C21H26N2O3
- Molecular Weight: 354.44
- MDL number: MFCD00005093
- EINECS: 205-672-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-11 20:10:50
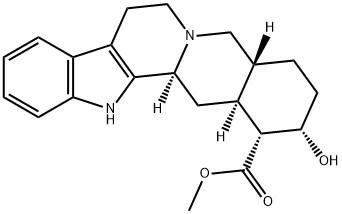
What is Yohimbine?
Absorption
Rapidly absorbed following oral administration. Bioavailability is highly variable, ranging from 7 to 87% (mean 33%).
Description
Yohimbine is a natural alkaloid. It was first extracted from the barks of Corynanthe yohimbe, a species of Rubiaceae trees in West Africa. It was reported that, in the dried bark of Pausinystalia johimbe, the content of mixed alkaloids is higher than 6.1%, in which the main component is yohimbine, indicating a great prospect for development . Yohimbe bark has been used as an aphrodisiac in Africa since ancient times. In 1900, it was applied by Kowit and Muller to patients with impotence and paralytic insensitivity caused by neurasthenia and obtained curative effect. From then on, clinical application of yohimbine began. Currently, yohimbine is a pure plant preparation in the treatment of erectile dysfunction with more affirmation and more applications.
Description
Yohimbine is an indole alkaloid ester first identified in several herbal species by the American botanist R. Raymond-Hamet in 1934. Today its main sources are the African evergreen yohimbe (Pausinystalia yohimbe) and Indian snakeroot (Rauwolfia serpentina). Yohimbe bark is sold over the counter as an herbal aphrodisiac. Yohimbine hydrochloride is a prescription drug that has been used to treat erectile dysfunction with varying results.
The Uses of Yohimbine
Yohimbine occurs in Corinanthe johimbeK. and Rubiaceae trees. It is also foundin the roots of Rauwolfia serpentina L.and Apocyanaceae. Its derivatives areused therapeutically as adrenergic blockingagents.
Background
A plant alkaloid with alpha-2-adrenergic blocking activity. Yohimbine has been used as a mydriatic and in the treatment of impotence. It is also alleged to be an aphrodisiac.
Indications
Indicated as a sympatholytic and mydriatic. Impotence has been successfully treated with yohimbine in male patients with vascular or diabetic origins and psychogenic origins.
Pharmacokinetics
Yohimbine is an indolalkylamine alkaloid with chemical similarity to reserpine. Yohimbine blocks presynaptic alpha-2 adrenergic receptors. Its action on peripheral blood vessels resembles that of reserpine, though it is weaker and of short duration. Yohimbine's peripheral autonomic nervous system effect is to increase parasympathetic (cholinergic) and decrease sympathetic (adrenergic) activity. It is to be noted that in male sexual performance, erection is linked to cholinergic activity and to alpha-2 adrenergic blockade which may theoretically result in increased penile inflow, decreased penile outflow or both. Yohimbine exerts a stimulating action on the mood and may increase anxiety. Such actions have not been adequately studied or related to dosage although they appear to require high doses of the drug. Yohimbine has a mild anti-diuretic action, probably via stimulation of hypothalmic center and release of posterior pituitary hormone. Reportedly Yohimbine exerts no significant influence on cardiac stimulation and other effects mediated by (beta)-adrenergic receptors. Its effect on blood pressure, if any, would be to lower it; however, no adequate studies are at hand to quantitate this effect in terms of Yohimbine dosage.
Metabolism
Yohimbine appears to undergo extensive metabolism in an organ of high flow such as the liver or kidney, however, the precise metabolic fate of yohimbine has not been fully determined.
Properties of Yohimbine
| Melting point: | 231-233 °C(lit.) |
| Boiling point: | 487.66°C (rough estimate) |
| Density | 1.1640 (rough estimate) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | THF; DMSO; Chloroform; |
| form | Solid |
| color | Beige |
Safety information for Yohimbine
Computed Descriptors for Yohimbine
Abamectin manufacturer
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran






You may like
-
 146-48-5 Yohimbine 98%View Details
146-48-5 Yohimbine 98%View Details
146-48-5 -
 146-48-5 98%View Details
146-48-5 98%View Details
146-48-5 -
 Yohimbine 99%View Details
Yohimbine 99%View Details
146-48-5 -
 Yohimbine 146-48-5 98%View Details
Yohimbine 146-48-5 98%View Details
146-48-5 -
 146-48-5 Yohimbine 99%View Details
146-48-5 Yohimbine 99%View Details
146-48-5 -
 143-07-7 99%View Details
143-07-7 99%View Details
143-07-7 -
 20776-67-4 99%View Details
20776-67-4 99%View Details
20776-67-4 -
 acid blue 113, acid navy blue , wool navy blue 0View Details
acid blue 113, acid navy blue , wool navy blue 0View Details
3351-05-1