Montelukast sodium
Synonym(s):1-[[[(1R)-1-[3-[(1E)-2-(7-Chloro-2-quinolinyl)ethenyl]phenyl]-3-[2-(1-hydroxy-1-methylethyl)phenyl]propyl]thio]methyl]cyclopropaneacetic acid sodium salt hydrate;Montelukast sodium
- CAS NO.:151767-02-1
- Empirical Formula: C35H37ClNNaO3S
- Molecular Weight: 610.18
- MDL number: MFCD00931431
- EINECS: 604-813-7
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-04-26 17:21:34
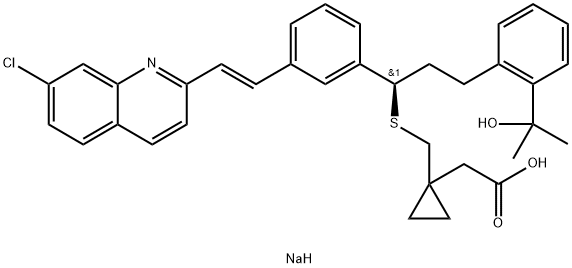
What is Montelukast sodium?
Description
Montelukast was launched as Singulair in Mexico and Finland for the management of mild to moderate asthma inadequately controlled by inhaled corticosteroids and short-acting beta2-agonists. Montelukast can be obtained by an seven-step synthesis from 3-[2(E)-(7-chloroquinolin-2-yl)vinyl] benzaldehyde. Montelukast is a potent, selective and orally active antagonist of the CysLT1 (formerly called LTD4) receptor, thus blocking the effects of the cysteinyl leukotrienes LTC4, LTD4 and LTE4 on microvascular permeability and the activation of eosinophils. Montelukast represents the third molecule of this class which has been approved in asthma after pranlukast (1995) and zafirlukast (1996). Montelukast has been studied extensively in placebo-controlled clinical trials, in mildly or severe asthmatic patients challenged with LTD4 or exercise. A variety of acute bronchoconstricting challenges were inhibited or attenuated with all doses used. Montelukast demonstrated clinically significant improvements in the parameters of asthma control associated with an appreciable improvement in quality of life., reducing days with asthma exacerbations and allowing significant tapering of corticosteroids. Montelukast is well-tolerated and only needs to be administered once a day.
The Uses of Montelukast sodium
Montelukast sodium hydrate has been used as a positive control drug to study the protective effects of Gumiganghwal-tang aqueous extract (GGTA) against airway inflammation and pulmonary fibrosis.
What are the applications of Application
Montelukast Sodium is a potent and highly selective CysLT1 receptor antagonist, without demonstrated CysLT2 activity
Properties of Montelukast sodium
| Melting point: | 115 °C(dec.) |
| storage temp. | 2-8°C |
| solubility | DMSO: ≥8mg/mL at 60°C |
| form | powder |
| color | white to tan |
Safety information for Montelukast sodium
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Health Hazard GHS08 |
| GHS Hazard Statements |
H318:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for Montelukast sodium
| InChIKey | LBFBRXGCXUHRJY-HKHDRNBDSA-M |
Abamectin manufacturer
Chynops Pharma
Dr. Reddy's Laboratories Ltd
SNECOFRi PVT LTD
SUPREME DRUGS PVT LTD
Vadivarhe Speciality Chemicals Limited
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran
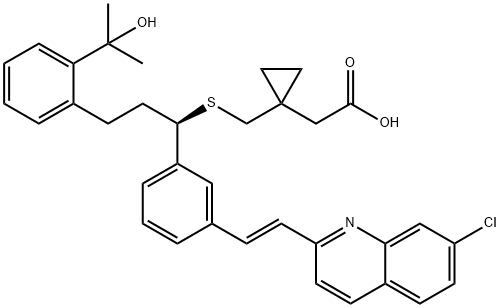

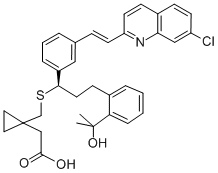
You may like
-
 151767-02-1 MONTELUKAST SODIUM 98%View Details
151767-02-1 MONTELUKAST SODIUM 98%View Details
151767-02-1 -
 Montelukast Sodium 98%View Details
Montelukast Sodium 98%View Details
151767-02-1 -
 Montelukast Sodium 15176-02-1 99%View Details
Montelukast Sodium 15176-02-1 99%View Details
15176-02-1 -
 Montelukast sodium 98%View Details
Montelukast sodium 98%View Details -
 151767-02-1 99%View Details
151767-02-1 99%View Details
151767-02-1 -
 151767-02-1 Montelukast sodium 98%View Details
151767-02-1 Montelukast sodium 98%View Details
151767-02-1 -
 151767-02-1 Montelukast sodium 99%View Details
151767-02-1 Montelukast sodium 99%View Details
151767-02-1 -
 Montelukast Sodium Ip 99%View Details
Montelukast Sodium Ip 99%View Details