Sotalol
- CAS NO.:3930-20-9
- Empirical Formula: C12H20N2O3S
- Molecular Weight: 272.36
- MDL number: MFCD00242936
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-03-19 15:37:50
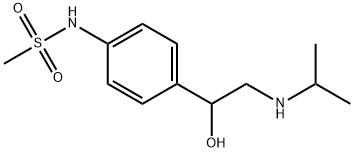
What is Sotalol?
Absorption
Sotalol is 90-100% bioavailable. When taken with a meal, adsorption is lowered by 18%. In patients with a creatinine clearance >80mL/min, the maximum concentration is 6.25±2.19.
Toxicity
Patients experiencing an overdose may present with bradycardia, congestive heart failure, hypotension, bronchospasm, and hypoglycemia. Larger intentional overdoses may present as hypotension, bradycardia, cardiac asystole, prolonged QT interval, torsade de pointes, ventricular tachycardia, and premature ventricular complexes. Stop administering sotalol and observe the patient until the QT interval returns to normal and the heart rate rises above 50 beats per minute. Hemodialysis may help lower plasma concentrations of sotalol as it is not bound to plasma proteins. Bradycardia and cardiac asystole may be treated with atropine, other anticholinergic drugs, beta adrenergic agonists, or transvenous cardiac pacing.. Second or third degree heart block may be treated with a transvenous cardiac pacemaker. Hypotension may be treated with epinephrine or norepinephrine. Bronchospasm may be treated with aminophylline or a beta-2 agonist, possibly at higher than normal doses. Torsade de pointes may be treated with DC cardioversion, transvenous cardiac pacing, epinephrine, or magnesium sulfate.
The oral LD50 for rats is 3450mg/kg, intraperitoneal LD50 for rats is 680mg/kg, oral LD50 for mice is 2600mg/kg, and intraperitoneal LD50 for mice is 670mg/kg.
Pregnant rabbits given 6 times the maximum recommended human dose showed an increase in fetal death and maternal toxicity, while rats given 18 times the maximum recommended human dose had an increased number of fetal resorptions. Sotalol is present in human breast milk so patients taking sotalol should not breast feed.
Sotalol has not been found to be carcinogenic. No studies have been performed regarding mutagenicity or clastogenicity. In animal studies, sotalol was not associated with a reduction in fertility aside from smaller litter sizes.
Further information regarding adverse reactions can be found here.
Indications
Sotalol is indicated to treat life threatening ventricular arrhytmias and maintain normal sinus rhythm in patients with atrial fibrillation or flutter. There are also oral solutions and intravenous injections indicated for patients requiring sotalol, but for whom a tablet would not be appropriate.
Background
Sotalol is a methanesulfonanilide developed in 1960. It was the first of the class III anti arrhythmic drugs. Sotalol was first approved as an oral tablet on 30 October 1992. A racemic mixture of sotalol is currently formulated as a tablet, oral solution, and intravenous injection indicated for life threatening ventricular arrhythmias and maintaining normal sinus rhythm in atrial fibrillation or flutter.
Pharmacokinetics
Sotalol is a competitive inhibitor of the rapid potassium channel. This inhibition lengthens the duration of action potentials and the refractory period in the atria and ventricles. The inhibition of rapid potassium channels is increases as heart rate decreases, which is why adverse effects like torsades de points is more likely to be seen at lower heart rates. L-sotalol also has beta adrenergic receptor blocking activity seen above plasma concentrations of 800ng/L. The beta blocking ability of sotalol further prolongs action potentials. D-sotalol does not have beta blocking activity but also reduces a patient's heart rate while standing or exercising. These actions combine to produce a negative inotropic effect that reduces the strength of contractility of muscle cells in the heart. Extension of the QT interval is also adversely associated with the induction of arrhythmia in patients.
Hyperglycemia is a greater risk for non insulin dependant diabetics than insulin dependant diabetics. Beta blockers inhibit insulin secretion which may cause hyperglycemia in type II diabetes mellitus. The risk of hypoglycemia is higher in insulin dependant diabetes than non insulin dependant diabetics. Beta blockers decrease secretion of insulin, which may mask hypoglycemia in an insulin dependant patient. Beta blockers also increase glucose uptake into cells which may prolong or potentiate hypoglycemia.
Further information regarding adverse reactions can be found here.
Metabolism
Sotalol is not metabolized.
Properties of Sotalol
| Melting point: | 206.5-207 °C |
| Boiling point: | 443.3±55.0 °C(Predicted) |
| Density | 1.298 g/cm3 |
| storage temp. | Hygroscopic, -20°C Freezer, Under inert atmosphere |
| solubility | Chloroform (Slightly, Heated), Methanol (Slightly, Sonicated) |
| form | Solid |
| color | Pale Yellow to Light Yellow |
Safety information for Sotalol
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H319:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Sotalol
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine N-Benzyl-3-hydroxypiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran

You may like
-
 2-Bromo-5-Iodopyridine 73290-22-9 98%View Details
2-Bromo-5-Iodopyridine 73290-22-9 98%View Details
73290-22-9 -
 141-86-6 98%View Details
141-86-6 98%View Details
141-86-6 -
 4-Amino-2-Chloropyridine 14432-12-3 98%View Details
4-Amino-2-Chloropyridine 14432-12-3 98%View Details
14432-12-3 -
 16867-03-1 2-Amino-3-Hydroxypyridine 98%View Details
16867-03-1 2-Amino-3-Hydroxypyridine 98%View Details
16867-03-1 -
 766-11-0 5-Bromo-2-Fluoropyridine 98%View Details
766-11-0 5-Bromo-2-Fluoropyridine 98%View Details
766-11-0 -
 2,3-Diamino-5-Chloropyridine 25710-20-7 98%View Details
2,3-Diamino-5-Chloropyridine 25710-20-7 98%View Details
25710-20-7 -
 13466-41-6 98%View Details
13466-41-6 98%View Details
13466-41-6 -
 Ammonium HexachloropalladateView Details
Ammonium HexachloropalladateView Details
19168-23-1