Sodium citrate
Synonym(s):Citric acid trisodium salt dihydrate;Sodium citrate solution;Sodium citrate tribasic dihydrate;Trisodium citrate dihydrate
- CAS NO.:68-04-2
- Empirical Formula: C6H9NaO7
- Molecular Weight: 216.12
- MDL number: MFCD00012462
- EINECS: 200-675-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-28 19:59:05
What is Sodium citrate?
Absorption
Tmax of 98-130min .
Toxicity
Overdose toxicity is mainly due to alkalosis as well as tetany or depressed heart function due to lack of free calcium .
Description
Sodium citrate may refer to any of the sodium salts of citric acid (though most commonly the third):
Mono sodium citrate
Di sodium citrate
Tri sodium citrate
As food additives, the 3 forms of the salt are also collectively known by the E number E331.
The Uses of Sodium citrate
sodium citrate may be used in cosmetic formulations as an alkalizer and to bind trace metals in solutions (by means of a chelating action). It is the sodium salt of citric acid.
What are the applications of Application
Citrate Concentrated Solution is 1M; may be used to prepare citrate buffer for antigen unmasking in IHC applications
What are the applications of Application
Citric Acid Trisodium Salt is used in the preparation of sodium citrate buffer for antigen unmasking in IHC
Indications
Used as an anticoagulant during plasmophoresis as well as a neutralizing agent in the treatment of upset stomach and acidic urine .
Background
Sodium citrate is the sodium salt of citric acid. It is white, crystalline powder or white, granular crystals, slightly deliquescent in moist air, freely soluble in water, practically insoluble in alcohol. Like citric acid, it has a sour taste. From the medical point of view, it is used as alkalinizing agent. It works by neutralizing excess acid in the blood and urine. It has been indicated for the treatment of metabolic acidosis.
Pharmacokinetics
Citrate prevents activation of the clotting cascade by chelating calcium ions. Citrate neutralizes acid in the stomach and urine, raising the pH .
Metabolism
Citrate is metabolized to bicarbonate in the liver and plays a role as an intermediate in the citric acid cycle .
Properties of Sodium citrate
| Melting point: | 300°C |
| Density | 1.008 g/mL at 20 °C |
| storage temp. | 2-8°C |
| solubility | Water (Slightly) |
| form | Liquid |
| color | White to off-white |
| Water Solubility | Soluble in water. |
| Sensitive | Hygroscopic |
Safety information for Sodium citrate
Computed Descriptors for Sodium citrate
| InChIKey | HRXKRNGNAMMEHJ-UHFFFAOYSA-K |
Abamectin manufacturer
Yogi Dye Chem Industries
Rishi Chemical Works Pvt Ltd
UNILOSA INTERNATINAL PRIVATE LIMITED
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine N-Benzyl-3-hydroxypiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran


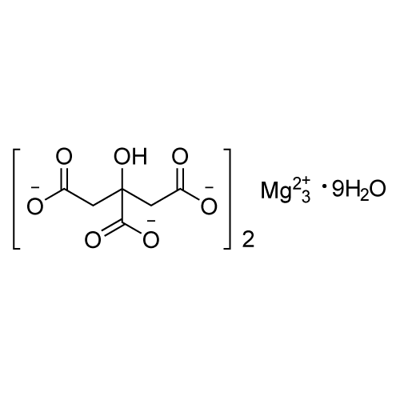
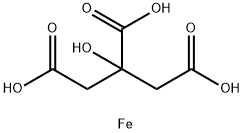
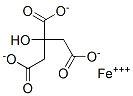
You may like
-
 Sodium Citrate 98%View Details
Sodium Citrate 98%View Details -
 SODIUM CITRATE 99%View Details
SODIUM CITRATE 99%View Details -
 Sodium Citrate 99%View Details
Sodium Citrate 99%View Details -
 Sodium Citrate 98%View Details
Sodium Citrate 98%View Details -
 Sodium Citrate 99%View Details
Sodium Citrate 99%View Details -
 Sodium citrate 99%View Details
Sodium citrate 99%View Details -
 Sodium citrate, tribasic 99%View Details
Sodium citrate, tribasic 99%View Details -
 Sodium citrate, tribasic 98%View Details
Sodium citrate, tribasic 98%View Details