Potassium hydroxide
Synonym(s):Potassium hydroxide;Caustic potash;Potassium hydroxide solution;Potassium hydroxide hydrate;caustic potash monohydrate
- CAS NO.:1310-58-3
- Empirical Formula: KOH
- Molecular Weight: 56.11
- MDL number: MFCD00003553
- EINECS: 215-181-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-04-29 22:41:02
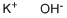
What is Potassium hydroxide ?
Absorption
KOH in aqueous solutions completely dissociates into K+ and OH- ions. Because of the neutralization of OH- by gastric HCl and the rapid blood pH regulation action (buffer capacity of extracellular body fluids, respiratory and renal compensation mechanisms), an alkalosis due to the OH- ions after KOH oral dosage in non-irritating conditions is thus prevented .
The uptake of potassium, in potassium hydroxide form, is much less than the oral uptake with therapeutic doses of KCl for treating potassium deficiency, of up to 10 g/day. Furthermore, the oral uptake of potassium from food/natural sources or from food additives is likely to be also much higher .
Toxicity
No studies are currently identified regarding the reproduction/developmental toxicity of potassium hydroxide .
The Ld50 of potassium hydroxide in rats ranges from 0.273 - 1.230 g KOH/kg body weight/day .
Adverse effects include vomiting, diarrhea, skin blistering, gastrointestinal disturbance, and burns .
Strong alkaline chemicals such as potassium hydroxide destroy soft tissues may cause a deep, penetrating type of burn. Caustics are usually hydroxides of light metals. Sodium hydroxide and potassium hydroxide are the most broadly used caustic agents in the industry .
Potassium Hydroxide can irritate the lungs. Repeated exposure may cause bronchitis to develop with coughing, phlegm, and/or shortness of breath .
Description
Potassium hydroxide (KOH) is a strong base commonly used in organic chemistry. It is typically bought in pellet form as 85% KOH, 15% water. The pKa of the conjugate acid of potassium hydroxide is 15.7. The hydroxide ion is not only a base, it is also an effective nucleophile. It is capable of nucleophilic reactions such as the diplacement of alkyl halides to form alcohols, and the hydrolysis of esters and nitriles to carboxylic acids.
The Uses of Potassium hydroxide
Potassium Hydroxide is a water-soluble food additive and bleaching agent. upon exposure to air it readily absorbs carbon dioxide and moisture and deliquesces. it is used to destroy the bitter chemical constituents in olives that will be used as black olives.
What are the applications of Application
Potassium hydroxide is buffer reagent
Indications
Medically, the microscopic examination of potassium hydroxide (KOH) preparations is utilized in the diagnosis of fungal hyphae or trichomonads .
Samples from hair, skin, or nail tissue are obtained by scraping with a scalpel, cotton-tipped applicator and are inoculated directly onto the KOH solution .
In addition to the above, potassium hydroxide is used as a softener for nail grooves .
Background
Potassium hydroxide, also known as lye is an inorganic compound with the chemical formula KOH. Also commonly referred to as caustic potash, it is a potent base that is marketed in several forms including pellets, flakes, and powders. It is used in various chemical, industrial and manufacturing applications. Potassium hydroxide is also a precursor to other potassium compounds. Potassium hydroxide is used in food to adjust pH, as a stabilizer, and as a thickening agent. This ingredient has been considered as generally safe as a direct human food ingredient by the FDA, based upon the observance of several good manufacturing practice conditions of use.
In addition to the above uses, potassium hydroxide is also used in making soap, as an electrolyte in alkaline batteries and in electroplating, lithography, and paint and varnish removers. Liquid drain cleaners contain 25 to 36% of potassium hydroxide .
Medically, potassium hydroxide (KOH) is widely used in the wet mount preparation of various clinical specimens for microscopic visualization of fungi and fungal elements in skin, hair, nails, and even vaginal secretions , .
Recently, it has been studied for efficacy and tolerability in the treatment of warts. It was determined that topical KOH solution was found to be a safe and effective treatment of plane warts .
Pharmacokinetics
The corrosiveness of potassium hydroxide renders it a very useful agent in the decomposition/removal soft tissue and hair removal. It is incorporated into some nail products, shaving creams, and soaps .
Metabolism
KOH in aqueous solution is entirely dissociated into K+ and OH- ions. Due to the neutralization of OH- by gastric HCl and the quick and efficient blood pH regulation mechanisms (buffer capacity of extra cellular body fluids, respiratory and renal compensation mechanisms), an alkalosis due to the OH- ions after KOH oral dosage in non-irritating conditions is prevented .
Properties of Potassium hydroxide
| Melting point: | 361 °C (lit.) |
| Boiling point: | 1320°C |
| Density | 1.450 g/mL at 20 °C |
| Flash point: | 52 °F |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | powder |
| appearance | White solid |
| color | white |
| Odor | Odorless |
| Water Solubility | soluble |
| Sensitive | Air Sensitive & Hygroscopic |
Safety information for Potassium hydroxide
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H290:Corrosive to Metals H302:Acute toxicity,oral H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P234:Keep only in original container. P260:Do not breathe dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Potassium hydroxide
| InChIKey | KWYUFKZDYYNOTN-UHFFFAOYSA-M |
Abamectin manufacturer
ANJI BIOSCIENCES
JSK Chemicals
Kronox Lab Sciences Pvt Ltd
Sree Rayalaseema HI Strength Hypo Ltd
Annexe Chem Pvt Ltd
HRV Global Life Sciences
Sujata Chemicals
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol 3-NITRO-5-ACETYL IMINODIBENZYL (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran

You may like
-
 Potassium Hydroxide supplierView Details
Potassium Hydroxide supplierView Details
1310-58-3 -
 Potassium hydroxide , 85% Pellets 99%View Details
Potassium hydroxide , 85% Pellets 99%View Details -
 Potassium hydroxide 98%View Details
Potassium hydroxide 98%View Details -
 1310-58-3 98%View Details
1310-58-3 98%View Details
1310-58-3 -
 Potassium hydroxide 98%View Details
Potassium hydroxide 98%View Details
1310-58-3 -
 Potassium hydroxide 98%View Details
Potassium hydroxide 98%View Details
1310-58-3 -
 Potassium hydroxide 1310-58-3 98%View Details
Potassium hydroxide 1310-58-3 98%View Details
1310-58-3 -
 Potassium hydroxide 1310-58-3 99%View Details
Potassium hydroxide 1310-58-3 99%View Details
1310-58-3