Phosphoric acid
Synonym(s):o-Phosphoric acid, Orthophosphoric acid;Orthophosphoric acid;Orthophosphoric acid, o-Phosphoric acid;Phosphoric acid;Phosphoric acid solution
- CAS NO.:7664-38-2
- Empirical Formula: H3O4P
- Molecular Weight: 98
- MDL number: MFCD00011340
- EINECS: 231-633-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-12-21 17:48:05
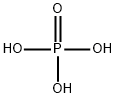
What is Phosphoric acid?
Absorption
Ortho phosphate is absorbed from, and to a limited extent secreted into, the gastrointestinal tract.
Toxicity
Man: LDLo, route unreported at 220mg/kg Rabbit: LD 50, Route: Skin, Reported dose: 2740 mg /kg, Effects: Behavioral : somnolence ( general depressed activity), Excitement Rat: LC50, Route: inhalation, Reported dose: >850 mg/m3/1h Rat: LD50, Route: oral, Reported dose: 1530 mg/kg, Effects: Kidney, ureter and bladder hematuria, Behavioral: somnolence (general depressed activity)
Description
Phosphoric acid was prepared first by Robert Boyle in 1694 by dissolving phosphorus pentoxide in water. Phosphoric acid is probably the most important compound of phosphorus. It is the second largest inorganic chemical by volume, after sulfuric acid, marketed in the United States.
The single most important application of Phosphoric acid is manufacturing phosphate salts for fertilizers. Such fertilizer phosphates include sodium, calcium, ammonium, and potassium phosphates. Other applications are in metal pickling and surface treatment for removal of metal oxides from metal surfaces; electropolishing of aluminum; as a bonding agent in various refractory products such as alumina and magnesia; as a catalyst in making nylon and gasoline; as a dehydrating agent; in fireproofing wood and fabrics; in lithographic engraving; in textile dyeing; in dental cement; in coagulating rubber latex; in purifying hydrogen peroxide; and as a laboratory reagent. Dilute solutions of phosphoric acid are used as additives to carbonated beverages for a pleasing sour taste. Also, dilute acid is used in refining sugar; as a nutrient; and as a buffering agent in preparing jam, jelly, and antibiotics. The commercial phosphoric acid is 85% (w/w) in strength.
Description
The most common form of phosphoric acid (H3PO4) is the 85% aqueous form. Pure anhydrous phosphoric acid is a white solid with a melting point of 42 C. Phosphoric acid has three pKa values covering a wide range (pKa's: 2.15, 7.20, 12.32). This is the reason why phosphoric acid solutions make a great choice as a buffer.
The Uses of Phosphoric acid
Phosphoric Acid is an acidulant that is an inorganic acid produced by burning phosphorus in an excess of air, producing phosphorus pentoxide which is dissolved in water to form orthophosphoric acid of varying concentrations. it is a strong acid which is soluble in water. the acid salts are termed phosphates. it is used as a flavoring acid in cola and root beer beverages to provide desirable acidity and sourness. it is used as a synergistic antioxidant in vegetable shorten- ings. in yeast manufacture, it is used to maintain the acidic ph and provide a source for phosphorus. it also functions as an acidulant in cheese. it is also termed orthophosphoric acid.
Indications
Phosphoric acid is used in dentistry and orthodontics as an etching solution, to clean and roughen the surfaces of teeth where dental appliances or fillings will be placed.
Background
Phosphoric Acid is a colorless, odorless phosphorus-containing inorganic acid. Phosphoric acid is a sequestering agent which binds many divalent cations, including Fe++, Cu++, Ca++, and Mg++.
What are the applications of Application
Phosphoric acid is an inorganic acid and phosphorylating agent
What are the applications of Application
Phosphoric acid solution is useful for HPLC
Pharmacokinetics
Transport of phosphate from the gut lumen is an active, energy-dependent process that is modified by several factors. ... Vitamin D stimulates phosphate absorption, an effect reported to precede its action on calcium ion transport.
Metabolism
Not Available
Properties of Phosphoric acid
| Melting point: | ~40 °C(lit.) |
| Boiling point: | 158 °C(lit.) |
| Density | 1.685 g/mL at 25 °C(lit.) |
| storage temp. | no restrictions. |
| solubility | H2O: soluble |
| form | Solid or Viscous Liquid |
| appearance | Colorless, viscous liquid (85% aq) |
| color | ≤10(APHA) |
| Odor | Odorless |
| Water Solubility | MISCIBLE |
Safety information for Phosphoric acid
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H290:Corrosive to Metals H302:Acute toxicity,oral H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P234:Keep only in original container. P270:Do not eat, drink or smoke when using this product. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Phosphoric acid
| InChIKey | NBIIXXVUZAFLBC-UHFFFAOYSA-N |
Abamectin manufacturer
ANJI BIOSCIENCES
SUKHA CHEMICAL INDUSTRIES
UNILOSA INTERNATINAL PRIVATE LIMITED
VITRAG CHEMICALS PVT LTD
JSK Chemicals
PUNJAB CHEMICALS AND CROP PROTECTION LTD
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol 3-NITRO-5-ACETYL IMINODIBENZYL (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran


You may like
-
 Phosphoric Acid 98%View Details
Phosphoric Acid 98%View Details -
 Phosphoric Acid 99%View Details
Phosphoric Acid 99%View Details -
 Phosphoric Acid 99%View Details
Phosphoric Acid 99%View Details -
 Phosphoric Acid 99%View Details
Phosphoric Acid 99%View Details
7664-38-2 -
 PHOSPHORIC ACID/ LIQUID 98%View Details
PHOSPHORIC ACID/ LIQUID 98%View Details -
 Phosphoric Acid (H3PO4) 99%View Details
Phosphoric Acid (H3PO4) 99%View Details -
 7664-38-2 98%View Details
7664-38-2 98%View Details
7664-38-2 -
 7664-38-2 Phosphoric acid 99%View Details
7664-38-2 Phosphoric acid 99%View Details
7664-38-2