Phenol
Synonym(s):Phenol;Phenol solution;Hydroxybenzene;C?H?O;Tris-equilabrated Phenol
- CAS NO.:108-95-2
- Empirical Formula: C6H6O
- Molecular Weight: 94.11
- MDL number: MFCD00002143
- EINECS: 203-632-7
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-27 17:30:58
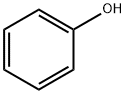
What is Phenol?
Description
Phenol is a stable chemical substance and appear as colourless/white crystals with a characteristic, distinct aromatic/acrid odour. It is reactive and incompatible with strong oxidising agents, strong bases, strong acids, alkalis, and calcium hypochlorite. Phenol is flammable and may discolour in light. Phenol is used in the manufacture or production of explosives, fertiliser, coke, illuminating gas, lampblack, paints, paint removers, rubber, perfumes, asbestos goods, wood preservatives, synthetic resins, textiles, drugs, and pharmaceutical preparations. It is also extensively used as a disinfectant in the petroleum, leather, paper, soap, toy, tanning, dye, and agricultural industries.
The Uses of Phenol
phenol is frequently used for medical chemical face peels. It may trap free radicals and can act as a preservative. Phenol, however, is an extremely caustic chemical with a toxicity potential. It is considered undesirable for use in cosmetics. even at low concentrations, it frequently causes skin irritation, swelling, and rashes.
Background
Phenol is an antiseptic and disinfectant. It is active against a wide range of micro-organisms including some fungi and viruses, but is only slowly effective against spores. Phenol has been used to disinfect skin and to relieve itching. Phenol is also used as an oral analgesic or anesthetic in products such as Chloraseptic to treat pharyngitis. Additionally, phenol and its related compounds are used in surgical ingrown toenail treatment, a process termed phenolization. Research indicates that parental exposure to phenol and its related compounds are positively associated with spontaneous abortion. During the second world war, phenol injections were used as a means of execution by the Nazis. Phenol is a toxic compound whose vapours are corrosive to the skin, eyes, and respiratory tract.
Indications
Phenol is primarily indicated for minor sore throat pain, sore mouth, minor mouth irritation, and pain associated with canker sores. Additionally, phenol is indicated in the treatment of focal spasticity.
Metabolism
Phenyl sulfate, phenyl glucuronide, quinol sulfate, and quinol glucuronide were detected in human beings as phenol metabolites.
Absorption
Phenol is rapidly absorbed through the skin and into the lungs.
Health hazards
Phenol and its vapors are corrosive to the eyes, skin, and respiratory tract. The corrosive effect on skin and mucous membranes is due to a protein-degenerating effect. Repeated or prolonged skin contact with phenol may cause dermatitis, and potentially second and third-degree burns. Inhalation of phenol vapor may cause lung edema. Phenol may adversely effect the central nervous system and heart. Long-term, or repeated exposure, to phenol may have harmful effects on the liver and kidneys.
Toxicity
Mouse, Subcutaneous, LD50: 0.3-0.35 g/kg. (Duplay and Cazin, 1891; Tollens, 1905).Rat, Subcutaneous, LD50: 0.45. (Deichmann and Witherup, 1944).Rat, Oral, LD50: 0.53. (Deichmann and Witherup, 1944).Rat, Oral, LD50: 0.65. (Flickinger, 1976).Rat, Cutaneous, LD50: 0.67. (Conning and Hayes, 1970).
Properties of Phenol
| Melting point: | 40-42 °C(lit.) |
| Boiling point: | 182 °C(lit.) |
| Density | 1.071 g/mL at 25 °C(lit.) |
| Flash point: | 175 °F |
| storage temp. | 2-8°C |
| solubility | H2O: 50 mg/mL at 20 °C, clear, colorless |
| form | liquid |
| color | faintly yellow |
| Odor | Sweet, medicinal odor detectable at 0.06 ppm |
| Water Solubility | 8 g/100 mL |
| FreezingPoint | 41℃ |
| Sensitive | Air & Light Sensitive |
Safety information for Phenol
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Skull and Crossbones Acute Toxicity GHS06  Health Hazard GHS08  Environment GHS09 |
| GHS Hazard Statements |
H314:Skin corrosion/irritation H341:Germ cell mutagenicity H373:Specific target organ toxicity, repeated exposure H411:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P260:Do not breathe dust/fume/gas/mist/vapours/spray. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Phenol
| InChIKey | ISWSIDIOOBJBQZ-UHFFFAOYSA-N |
Abamectin manufacturer
SHAKAMBARI AROMATICS PRIVATE LIMITED
UNILOSA INTERNATINAL PRIVATE LIMITED
Aero Agro Chemicals Industries Ltd
KARPSCHEM LABORATORIES PVT. LTD.
JSK Chemicals
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran

You may like
-
 Phenol 99%View Details
Phenol 99%View Details -
 Phenol 99%View Details
Phenol 99%View Details -
 108-95-2 99%View Details
108-95-2 99%View Details
108-95-2 -
 Phenol 99%View Details
Phenol 99%View Details -
 108-95-2 98%View Details
108-95-2 98%View Details
108-95-2 -
 Phenol 108-95-2 98%View Details
Phenol 108-95-2 98%View Details
108-95-2 -
 108-95-2 Phenol 99%View Details
108-95-2 Phenol 99%View Details
108-95-2 -
 Phenol 99%View Details
Phenol 99%View Details
108-95-2