Maraviroc
Synonym(s):4,4-Difluoro-N-[(1S)-3-[(3-exo)-3-[3-methyl-5-(1-methylethyl)-4H-1,2,4-triazol-4-yl]-8-azabicyclo[3.2.1]oct-8-yl]-1-phenylpropyl]cyclohexanecarboxamide;UK-427857
- CAS NO.:376348-65-1
- Empirical Formula: C29H41F2N5O
- Molecular Weight: 513.68
- MDL number: MFCD09953791
- EINECS: 609-456-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-04-22 18:05:02
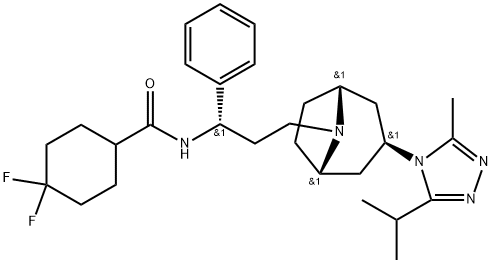
What is Maraviroc?
Absorption
The absolute oral bioavailability of a 100 mg dose is 23% and is predicted to be 33% at 300 mg. Coadministration of a 300mg tablet with a high fat breakfast reduced maraviroc Cmax and AUC by 33% in healthy volunteers.
Description
Maraviroc is the first CCR5 receptor antagonist that has been developed and launched for the treatment of HIV-1. Maraviroc binds in a slowly reversible, allosteric manner to CCR5, which is one of two principle chemokine co-receptors for viral entry into the host cell, the other being CXCR4. Binding of maraviroc to CCR5 induces conformational changes within the chemokine receptor, thereby preventing CCR5 binding to the viral gp120 protein and the ultimate CCR5- mediated virus-cell fusion that is a prerequisite for HIV invasion. Maraviroc, with its unique mechanism of action as a fusion inhibitor, joins the greater than 20 marketed antiretrovirals, including nucleotide reverse transcriptase inhibitors (NRTIs), non-nucleoside reverse transcriptase inhibitors (NNRTIs), and PIs. It is approved for use in combination with these other antiretroviral drugs in adult patients with R5-tropic HIV-1 infection (but not X4 or dual/mixed tropic HIV-1).
The Uses of Maraviroc
Maraviroc is a CCR5 antagonist for MIP-1α, MIP-1β and RANTES with IC50 of 3.3 nM, 7.2 nM and 5.2 nM, respectively
Indications
Maraviroc is indicated in combination with other antiretroviral agents for the treatment of CCR5-tropic HIV-1 infection in adults and pediatric patients weighing at least 2kg. It is not recommended in patients with dual/mixed- or CXCR4-tropic HIV-1.
Background
Maraviroc (brand-named Selzentry, or Celsentri outside the U.S.) is a chemokine receptor antagonist drug developed by the drug company Pfizer that is designed to act against HIV by interfering with the interaction between HIV and CCR5. It was originally labelled as UK-427857 during development but was assigned the Maraviroc name as it entered trials. It was approved for use by the FDA in August, 2007.
Pharmacokinetics
Maraviroc is a chemokine receptor antagonist drug developed by the drug company Pfizer that is designed to act against HIV by interfering with the interaction between HIV and CCR5.
Metabolism
In vitro studies indicate that CYP3A is the major enzyme responsible for maraviroc metabolism.
Properties of Maraviroc
| Melting point: | 79-81°C |
| Density | 1.29±0.1 g/cm3(Predicted) |
| storage temp. | room temp |
| solubility | DMSO: >30mg/mL |
| form | white powder |
| color | White |
Safety information for Maraviroc
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Maraviroc
Abamectin manufacturer
Aurona Med Pharma Private Limited
Cohance Lifesciences (Previously RA Chem Pharma Ltd)
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol 3-NITRO-5-ACETYL IMINODIBENZYL (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran


You may like
-
 376348-65-1 Maraviroc 98%View Details
376348-65-1 Maraviroc 98%View Details
376348-65-1 -
 376348-65-1 98%View Details
376348-65-1 98%View Details
376348-65-1 -
 Maraviroc 98%View Details
Maraviroc 98%View Details
376348-65-1 -
 Maraviroc 376348-65-1 98%View Details
Maraviroc 376348-65-1 98%View Details
376348-65-1 -
 68915-31-1 99%View Details
68915-31-1 99%View Details
68915-31-1 -
 Azadirachtin 11141-17-6 99%View Details
Azadirachtin 11141-17-6 99%View Details
11141-17-6 -
 Geraniol 99%View Details
Geraniol 99%View Details
106-24-1 -
 BENZALKONIUM CHLORIDE BKC 99%View Details
BENZALKONIUM CHLORIDE BKC 99%View Details
8001-54-5