L(+)-Arginine
Synonym(s):L-Arginine;ARG;(S)-2-Amino-5-guanidinopentanoic acid;Abelson murine leukemia viral oncogene homolog 2;Abelson tyrosine-protein kinase 2
- CAS NO.:74-79-3
- Empirical Formula: C6H14N4O2
- Molecular Weight: 174.2
- MDL number: MFCD00002635
- EINECS: 200-811-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-10 16:01:40
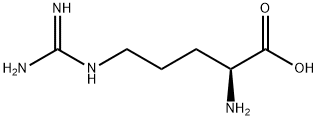
What is L(+)-Arginine?
Description
L-Glutamine and?L-arginine are classified as semi-essential or conditionally essential amino acids, which means that the human body can synthesize them under some health conditions but not others. Both are abundant in many foods.
Neither?D-glutamine nor?D-arginine is common in nature, so the?L-amino acids are usually referred to without prefixes. In 1883, German chemists Ernst. Schulze and E. Bosshard isolated?L-glutamine from the juice of sugarbeets. It is the most common amino acid in human blood and a key component of proteins.
In 1886, Schulze was also the first to isolate?L-arginine, in this case from lupin seedlings. It is also an important amino acid in protein biosynthesis.
It turns out that a difference in occurrence between these two amino acids makes a huge difference in huge animals. This year, while examining the melanocortin system* of cetaceans, Roger D. Cone at the University of Michigan (Ann Arbor) and colleagues discovered that the?two suborders of the sea mammals have different components at position 156 in the melanocortin-4 receptor. Glutamine is at this position in baleen whales, whereas toothed whales and dolphins contain arginine instead.
Cone’s group found that arginine in the receptor suppresses the beasts’ appetites relative to the glutamine-containing receptor. They believe that this difference contributes to the much larger sizes of baleen whales.
*The melanocortin system is an important regulator of metabolism, food intake, and fat storage.
Description
L-Glutamine and?L-arginine are classified as semi-essential or conditionally essential amino acids, which means that the human body can synthesize them under some health conditions but not others. Both are abundant in many foods.
Neither?D-glutamine nor?D-arginine is common in nature, so the?L-amino acids are usually referred to without prefixes. In 1883, German chemists Ernst. Schulze and E. Bosshard isolated?L-glutamine from the juice of sugarbeets. It is the most common amino acid in human blood and a key component of proteins.
In 1886, Schulze was also the first to isolate?L-arginine, in this case from lupin seedlings. It is also an important amino acid in protein biosynthesis.
It turns out that a difference in occurrence between these two amino acids makes a huge difference in huge animals. This year, while examining the melanocortin system* of cetaceans, Roger D. Cone at the University of Michigan (Ann Arbor) and colleagues discovered that the?two suborders of the sea mammals have different components at position 156 in the melanocortin-4 receptor. Glutamine is at this position in baleen whales, whereas toothed whales and dolphins contain arginine instead.
Cone’s group found that arginine in the receptor suppresses the beasts’ appetites relative to the glutamine-containing receptor. They believe that this difference contributes to the much larger sizes of baleen whales.
*The melanocortin system is an important regulator of metabolism, food intake, and fat storage.
The Uses of L(+)-Arginine
Amino acid; nutrient.
What are the applications of Application
L-Arginine is a precursor of nitric oxide synthesis that induces vasodilation
Metabolism
Not Available
Properties of L(+)-Arginine
| Melting point: | 222 °C (dec.) (lit.) |
| Boiling point: | 305.18°C (rough estimate) |
| Density | 1.2297 (rough estimate) |
| storage temp. | 2-8°C |
| solubility | H2O: 100 mg/mL |
| form | powder |
| appearance | white crystals or powder |
| appearance | white crystals or powder |
| color | white |
| Water Solubility | 148.7 g/L (20 ºC) |
| Sensitive | Air Sensitive |
Safety information for L(+)-Arginine
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H319:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P337+P313:IF eye irritation persists: Get medical advice/attention. P403:Store in a well-ventilated place. |
Computed Descriptors for L(+)-Arginine
| InChIKey | ODKSFYDXXFIFQN-BYPYZUCNSA-N |
Abamectin manufacturer
ADVENT CHEMBIO PRIVATE LIMITED
JSK Chemicals
HRV Global Life Sciences
Innovative Health Care (India) Pvt. Ltd.
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol 3-NITRO-5-ACETYL IMINODIBENZYL (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran
You may like
-
 74-79-3 L-Arginine, 98% 98%View Details
74-79-3 L-Arginine, 98% 98%View Details
74-79-3 -
 74-79-3 99%View Details
74-79-3 99%View Details
74-79-3 -
 L-Arginine, 99% 99%View Details
L-Arginine, 99% 99%View Details
74-79-3 -
 L(+)-Arginine 99%View Details
L(+)-Arginine 99%View Details -
 74-79-3 L(+)-Arginine 99%View Details
74-79-3 L(+)-Arginine 99%View Details
74-79-3 -
 74-79-3 98%View Details
74-79-3 98%View Details
74-79-3 -
 L(+)-Arginine 98%View Details
L(+)-Arginine 98%View Details
74-79-3 -
 Etoricoxib 98%View Details
Etoricoxib 98%View Details
202409-33-4