Ifosfamide
Synonym(s):Ifex;Ifosfamide;N,3-Bis(2-chloroethyl)tetrahydro-2H-1,3,2-oxazaphosphorin-2-amine-2-oxide
- CAS NO.:3778-73-2
- Empirical Formula: C7H15Cl2N2O2P
- Molecular Weight: 261.09
- MDL number: MFCD00057374
- EINECS: 223-237-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-06-08 09:02:20
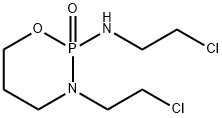
What is Ifosfamide?
Toxicity
LD50 (mouse) = 390-1005 mg/kg, LD50 (rat) = 150-190 mg/kg. Side effects include nausea, vomiting and myelosuppression. Toxic effects include central nervous system toxicity (confusion, hallucinations) and urotoxic effects (cystitis, blood in urine).
The Uses of Ifosfamide
A cytostatic agent, related structurally to cyclophosphamide
Background
Ifosfamide is a chemotherapeutic agent chemically related to the nitrogen mustards and a synthetic analog of cyclophosphamide. It is active as an alkylating agent and an immunosuppressive agent.
Indications
Used as a component of various chemotherapeutic regimens as third-line therapy for recurrent or refractory germ cell testicular cancer. Also used as a component of various chemotherapeutic regimens for the treatment of cervical cancer, as well as in conjunction with surgery and/or radiation therapy in the treatment of various soft tissue sarcomas. Other indications include treatment of osteosarcoma, bladder cancer, ovarian cancer. small cell lung cancer, and non-Hodgkin's lymphoma.
What are the applications of Application
Ifosfamide is a cytostatic agent structurally related to cyclophosphamide
Pharmacokinetics
Ifosfamide requires activation by microsomal liver enzymes to active metabolites in order to exert its cytotoxic effects. Activation occurs by hydroxylation at the ring carbon atom 4 to form the unstable intermediate 4-hydroxyifosfamide. This metabolite than rapidly degrades to the stable urinary metabolite 4-ketoifosfamide. The stable urinary metabolite, 4-carboxyifosfamide, is formed upon opening of the ring. These urinary metabolites have not been found to be cytotoxic. N, N-bis (2-chloroethyl)-phosphoric acid diamide (ifosphoramide) and acrolein are also found. The major urinary metabolites, dechloroethyl ifosfamide and dechloroethyl cyclophosphamide, are formed upon enzymatic oxidation of the chloroethyl side chains and subsequent dealkylation. It is the alkylated metabolites of ifosfamide that have been shown to interact with DNA. Ifosfamide is cycle-phase nonspecific.
Metabolism
Primarily hepatic. Ifosfamide is metabolized through two metabolic pathways: ring oxidation ("activation") to form the active metabolite, 4-hydroxy-ifosfamide and side-chain oxidation to form the inactive metabolites, 3-dechloro-ethylifosfamide or 2-dechloroethylifosfamide with liberation of the toxic metabolite, chloroacetaldehyde. Small quantities (nmol/mL) of ifosfamide mustard and 4-hydroxyifosfamide are detectable in human plasma. Metabolism of ifosfamide is required for the generation of the biologically active species and while metabolism is extensive, it is also quite variable among patients.
Properties of Ifosfamide
| Melting point: | 48°C |
| Boiling point: | 336.1±52.0 °C(Predicted) |
| Density | 1.33±0.1 g/cm3(Predicted) |
| storage temp. | Inert atmosphere,2-8°C |
| form | neat |
Safety information for Ifosfamide
| Signal word | Danger |
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06  Health Hazard GHS08 |
| GHS Hazard Statements |
H301:Acute toxicity,oral H319:Serious eye damage/eye irritation H340:Germ cell mutagenicity H350:Carcinogenicity H360:Reproductive toxicity |
| Precautionary Statement Codes |
P201:Obtain special instructions before use. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Ifosfamide
Abamectin manufacturer
KROMOZOME
Aarti Industries Limited (AIL)
Basil Drugs AND Pharmaceuticals Pvt Ltd
Aspen Biopharma Labs Pvt Ltd
Kekule Life Sciences Limited
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine N-Benzyl-3-hydroxypiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran
You may like
-
 Ifosfamide 98%View Details
Ifosfamide 98%View Details
3778-73-2 -
 Ifosfamide sterile 98%View Details
Ifosfamide sterile 98%View Details -
 3778-73-2 Ifosfamide 98%View Details
3778-73-2 Ifosfamide 98%View Details
3778-73-2 -
 3778-73-2 98%View Details
3778-73-2 98%View Details
3778-73-2 -
 Ifosfamide 98%View Details
Ifosfamide 98%View Details
3778-73-2 -
 Ifosfamide 3778-73-2 98%View Details
Ifosfamide 3778-73-2 98%View Details
3778-73-2 -
 Ifosfamide 3778-73-2 98%View Details
Ifosfamide 3778-73-2 98%View Details
3778-73-2 -
 Ifosfamide 3778-73-2 99%View Details
Ifosfamide 3778-73-2 99%View Details
3778-73-2