Dronedarone
- CAS NO.:141626-36-0
- Empirical Formula: C31H44N2O5S
- Molecular Weight: 556.76
- MDL number: MFCD00910331
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-18 11:31:13
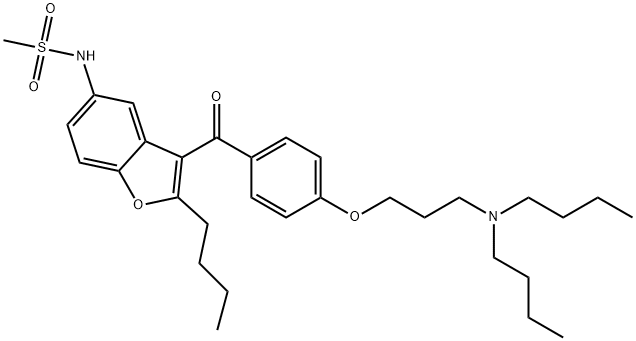
What is Dronedarone?
Absorption
Dronedarone is well absorbed after oral administration (>70%). It displays low systemic bioavailability due to extensive first-pass metabolism. The absolute bioavailability of dronedarone without and with a high-fat meal is 4% and 15%, respectively. The peak plasma concentrations of dronedarone and its main circulating N-debutyl metabolite are reached within 3 to 6 hours after administration with food.
Following repeated administration of 400 mg dronedarone twice daily, the steady-state was reached within 4 to 8 days of initial treatment. The steady-state Cmax and systemic exposure to the N-debutyl metabolite are similar to that of the parent compound.
Toxicity
In an acute toxicity study, the oral LD50 in rat was >2,000 mg/kg.
In oral studies, dronedarone showed a limited potential for toxicity in humans in acute overdose situations. However, it is recommended that the patient's cardiac rhythm and blood pressure is monitored in the event of overdose. Symptomatic and supportive treatments should be initiated.
Description
AF is the most common form of sustained cardiac arrhythmia, with
an increasing prevalence in the aging population. AF accounts for
34.5% of arrhythmia-related hospital admissions in the United States.
The most significant consequences of AF include congestive heart failure, a 5-fold increased risk of stroke, and increased rate of mortality.
Although a 90% conversion rate from AF to normal sinus rhythm (NSR)
can be achieved with electrical cardioversion, up to 70% of these
patients require additional therapy with antiarrhythmic drugs in order
to maintain NSR.
Dronedarone, a close
analog of amiodarone, is structurally modified to provide improved
safety and pharmacokinetic profile. With the introduction of a sulfonamide group, dronedarone is less lipophilic, has lower tissue accumulation, and has a much shorter serum half-life (~24 h) compared with
amiodarone. Additionally, dronedarone lacks the iodine moieties that
are responsible for thyroid dysfunctions associated with amiodarone.
Dronedarone is specifically indicated to reduce the risk of cardiovascular hospitalization in patients with paroxysmal or persistent AF or AFL,
with a recent episode of AF/AFL and associated cardiovascular risk
factors, who are in sinus rhythm or who will be cardioverted. Similar to
amiodarone, dronedarone is a potent blocker of multiple ion currents
(including the rapidly activating delayed-rectifier potassium current,
the slowly activating delayed-rectifier potassium current, the inward rectifier potassium current, the acetylcholine-activated potassium current, peak sodium current, and L-type calcium current) and exhibits
antiadrenergic effects. Overall, dronedarone was well tolerated. The
most common side effects were gastrointestinal in nature and
included nausea, vomiting, and diarrhea.
Background
Dronedarone is a Class III antiarrhythmic drug that works to restore the normal sinus rhythm in patients with paroxysmal or persistent atrial fibrillation. Atrial fibrillation is a common sustained arrhythmia where the treatment primarily focuses on stroke prevention and symptom management. It is managed by rate control, rhythm control, prevention of thromboembolic events, and treatment of the underlying disease. Similar to amiodarone, dronedarone is a multichannel blocker that works to control rhythm and rate in atrial fibrillation. It meets criteria of all four Vaughan Williams antiarrhythmic drug classes by blocking sodium, potassium, and calcium ion channels and inhibiting β-adrenergic receptors.
Dronedarone is a related benzofuran compound to amiodarone but its chemical structure lacks iodine moieties which are associated with amiodarone-induced thyroid problems. Additionally, the methyl sulfonyl group in its structure renders dronedarone to be more lipophilic with a shorter half-life than amiodarone. This ultimately leads to reduced tissue accumulation of the drug and decreased risk for organ toxicities, such as thyroid and pulmonary toxicities. Commonly marketed as Multaq?, dronedarone was approved by the FDA in July 2009 and Health Canada in August 2009. A safety concern for the risk of drug-induced hepatocellular injury has been issued following marketing of dronedarone.
Indications
Dronedarone is indicated for the management of atrial fibrillation (AF) in patients in sinus rhythm with a history of paroxysmal or persistent AF to reduce the risk of hospitalization.
Pharmacokinetics
Dronedarone is an antiarrhythmic agent that restores normal sinus rhythm and reduces heart rate in atrial fibrillation. In another model, it prevents ventricular tachycardia and ventricular fibrillation. Dronedarone moderately prolongs the QTc interval by about 10 ms on average. Dronedarone decreases arterial blood pressure and reduces oxygen consumption. It reduces myocardial contractility with no change in left ventricular ejection fraction. Dronedarone vasodilates coronary arteries through activation of the nitric oxide pathway. In clinical studies, dronedarone reduced incidence of hospitalizations for acute coronary syndromes and reduced incidence of stroke. Dronedarone exhibits antiadrenergic effects by reducing alpha-adrenergic blood pressure response to epinephrine and beta 1 and beta 2 responses to isoproterenol.
Dronedarone was shown to inhibit triiodothyronine (T3) signalling by binding to TRα1 but much less so to TRβ1. The treatment of dronedarone in patients with severe heart failure and left ventricular systolic dysfunction was associated with increased early mortality related to the worsening of heart failure. In animal studies, the use of dronedarone at doses equivalent to the recommended human doses was associated with fetal harm. In clinical studies and postmarketing reports, dronedarone was shown to cause hepatocellular liver injury and pulmonary toxicities, such as interstitial lung disease, pneumonitis, and pulmonary fibrosis. Compared to its related compound amiodarone, dronedarone has a faster onset and offset of actions with a shorter elimination half-life and low tissue accumulation.
Metabolism
Dronedarone predominantly undergoes CYP3A-mediated hepatic metabolism. Initial metabolism of dronedarone involves N-debutylation to form the N-debutyl-dronedarone, which retains 1/10 to 1/3 of pharmacological activity of the parent compound. N-debutyl-dronedarone can be further metabolized to phenol-dronedarone via O-dealkylation and propanoic acid-dronedarone via oxidative deamination. Dronedarone can also be metabolized by CYP2D6 to form benzofuran-hydroxyl-dronedarone. Other detectable metabolites include C-dealkyl-dronedarone and dibutylamine-hydroxyl-dronedarone, along with other minor downstream metabolites with undetermined chemical structures.
Properties of Dronedarone
| Melting point: | 65.3° |
| Boiling point: | 683.9±65.0 °C(Predicted) |
| Density | 1.143±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | ≥27.84 mg/mL in DMSO; insoluble in H2O; ≥49.8 mg/mL in EtOH |
| form | solid |
Safety information for Dronedarone
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H332:Acute toxicity,inhalation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Dronedarone
Abamectin manufacturer
Ralington Pharma
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol 3-NITRO-5-ACETYL IMINODIBENZYL (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran
You may like
-
 Dronedarone 98%View Details
Dronedarone 98%View Details -
 141626-36-0 98%View Details
141626-36-0 98%View Details
141626-36-0 -
 Dronedarone 98%View Details
Dronedarone 98%View Details
141626-36-0 -
 Dronedarone 141626-36-0 98%View Details
Dronedarone 141626-36-0 98%View Details
141626-36-0 -
 Dronedarone 141626-36-0 99%View Details
Dronedarone 141626-36-0 99%View Details
141626-36-0 -
 68915-31-1 99%View Details
68915-31-1 99%View Details
68915-31-1 -
 Geraniol 99%View Details
Geraniol 99%View Details
106-24-1 -
 BENZALKONIUM CHLORIDE BKC 99%View Details
BENZALKONIUM CHLORIDE BKC 99%View Details
8001-54-5