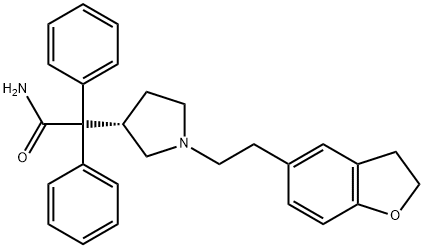
Darifenacin
- CAS NO.:133099-04-4
- Empirical Formula: C28H30N2O2
- Molecular Weight: 426.55
- MDL number: MFCD00896313
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-18 11:31:10
What is Darifenacin?
Absorption
The mean oral bioavailability at steady state is estimated to be 15% and 19% for 7.5 mg and 15 mg tablets, respectively.
Toxicity
Overdosage can potentially result in severe central anticholinergic effects.
Description
Darifenacin is a novel muscarinic M3 selective antagonist for the once-daily oral treatment of urinary incontinence and overactive bladder. The majority of overactive bladder symptoms are thought to result from the overactivity of the detrusor muscle, which is primarily mediated by acetylcholine-induced stimulation of muscarinic M3 receptors in the bladder. Consequently, antimuscarinic agents have become the mainstay of overactive bladder treatment. Darifenacin has a higher level of M3 selectivity than the previously marketed antimuscarinic agents. It has Ki values of 16nM for M1, 50 nM for M2, and 1.6 nM for M3 receptors. It is slightly more M3 selective than solifenacin (M1:Ki=25 nM, M2:Ki=126 nM, M3:Ki=10 nM), which was launched in 2004. Darifenacin is significantly more selective than other muscarinics such as tolterodine, oxybutynin, and trospium, which are all essentially equipotent against M1, M2, and M3 receptors. In addition,darifenacin demonstrates greater effect on tissues in which the predominant receptor type is M3 rather than M1 or M2. In vitro darifenacin inhibits carbacholinduced contractions with greater potency in isolated guinea-pig bladder (M3) than in guinea-pig atria (M2) or dog saphenous vein (M1). In animal models, it shows greater selectivity for inhibition of detrusor contraction over salivation or tachycardia.Darifenacin is supplied as a controlled release formulation, and the recommended dosage is 7.5 mg once, daily. Darifenacin is rapidly and completely absorbed from the GI tract after oral administration, with maximum plasma levels achieved after about 7 h. The elimination half-life is approximately 3 h, but because of the controlled release characteristics of the formulation, the drug is suitable for once-daily dosing. Steady-state plasma levels are achieved within 6 days of commencing treatment. Darifenacin exhibits high-protein binding (98%), a volume of distribution of 163 L, and a clearance of 40 L/h. It has low oral bioavailability (15–19%) due to extensive first-pass metabolism by CYP3A4 and CYP2D6, but this can be saturated after multiple administrations. The major circulating metabolites are produced by monohydroxylation and N-dealkylation; however, none contribute significantly to the overall clinical effect of darifenacin. Approximately 58% of the dose is excreted in urine and 44% in feces; only a small percentage (3%) of the excreted dose is unchanged darifenacin.
The Uses of Darifenacin
Treatment for an overactive bladder.
Indications
For the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency and frequency.
Background
Darifenacin (Enablex?, Novartis) is a medication used to treat urinary incontinence.
Darifenacin blocks M3 muscarinic acetylcholine receptors, which mediate bladder muscle contractions. This block reduces the urgency to urinate and so it should not be used in people with urinary retention.
It is unknown if M3 receptor selectivity is clinically advantageous in overactive bladder syndrome treatments.
Pharmacokinetics
Darifenacin is a competitive muscarinic receptor antagonist. In vitro studies using human recombinant muscarinic receptor subtypes show that darifenacin has greater affinity for the M3 receptor than for the other known muscarinic receptors (9 and 12-fold greater affinity for M3 compared to M1 and M5, respectively, and 59-fold greater affinity for M3 compared to both M2 and M4). Muscarinic receptors play an important role in several major cholinergically mediated functions, including contractions of the urinary bladder smooth muscle and stimulation of salivary secretion. Adverse drug effects such as dry mouth, constipation and abnormal vision may be mediated through effects on M3 receptors in these organs.
Metabolism
Hepatic. Primarily mediated by the cytochrome P450 enzymes CYP2D6 and CYP3A4.
Properties of Darifenacin
| Boiling point: | 614.3±55.0 °C(Predicted) |
| Density | 1.192±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | Soluble in DMSO |
| form | Powder |
Safety information for Darifenacin
Computed Descriptors for Darifenacin
Abamectin manufacturer
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol 3-NITRO-5-ACETYL IMINODIBENZYL (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran



You may like
-
 133099-04-4 Darifenacin 98%View Details
133099-04-4 Darifenacin 98%View Details
133099-04-4 -
 133099-04-4 99%View Details
133099-04-4 99%View Details
133099-04-4 -
 Darifenacin 99%View Details
Darifenacin 99%View Details -
 68915-31-1 99%View Details
68915-31-1 99%View Details
68915-31-1 -
 Azadirachtin 11141-17-6 99%View Details
Azadirachtin 11141-17-6 99%View Details
11141-17-6 -
 Geraniol 99%View Details
Geraniol 99%View Details
106-24-1 -
 BENZALKONIUM CHLORIDE BKC 99%View Details
BENZALKONIUM CHLORIDE BKC 99%View Details
8001-54-5 -
 Amrit Neem 11141-17-6 99%View Details
Amrit Neem 11141-17-6 99%View Details
11141-17-6