CARBON MONOXIDE
- CAS NO.:630-08-0
- Empirical Formula: CO
- Molecular Weight: 28.01
- MDL number: MFCD00011320
- EINECS: 211-128-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-03-14 15:18:25
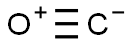
What is CARBON MONOXIDE?
Absorption
Although CO is not one of the respiratory gases, the similarity of physico-chemical properties of CO and oxygen (O2) permits an extension of the findings of studies on the kinetics of transport of O2 to those of CO. The rate of formation and elimination of COHb, its concentration in blood, and its catabolism is controlled by numerous physical factors and physiological mechanisms .
The absorption of carbon monoxide from the consumption treated food products is not significant. Risk of CO toxicity from the packaging process or from consumption of CO-treated meats is negligible .
Toxicity
LD50 is 1807 ppm in rats after 4 hours of exposure . In humans, exposure to 4000 ppm or more in less than 1 hour leads to death .
Carbon Monoxide Toxicity from Food Ingestion Treated with Carbon Monoxide
Very little information has been published in the literature on the consumer’s exposure to CO-packaged meat. The toxicological aspects of CO used in MAP of meat were reviewed by S?rheim et al. , and they concluded that, with up to about 0.5% of CO, no human toxicity is likely. It has been suggested that the consumption of CO-treated meat is not associated with any health risks, and meat from CO-MAP results only in negligible amounts of CO and COHb in humans , .
Mechanism of CO Toxicity
Once CO is inhaled, it binds with hemoglobin to form carboxyhemoglobin (COHb) with an affinity of 200 times greater than oxygen that leads to decreased oxygen-carrying capacity and the decreased release of oxygen to tissues, causing tissue hypoxia. Ischemia occurs with CO poisoning when there is a loss of consciousness combined with hypotension and ischemia in the arterial border areas of the brain. Besides binding to many heme-containing proteins, CO interrupts oxidative metabolism, leading to the formation of free radicals. Once hypotension and unconsciousness occur with CO poisoning, lipid peroxidation and apoptosis soon follow .
Failure to diagnose CO poisoning may result in morbidity and mortality and allow for continued exposure to a dangerous environment. The management of CO poisoning begins with inhalation of supplemental oxygen therapy and aggressive supportive measures. Hyperbaric oxygen therapy (HBOT) accelerates the dissociation of CO from hemoglobin .
The concentration, exposure time and physical activity of the individual will determine the percentage conversion of haemoglobin to carboxyhaemoglobin. The effects produced depend on the degree and duration of saturation of blood with carbon monoxide .
Levels of Intoxication with Carbon Monoxide
The first symptoms of carbon monoxide exposure when carboxyhemoglobin is 15-30% are generalized, and may include: headache, dizziness, nausea, fatigue, and impaired manual dexterity. Individuals with ischemic heart disease may suffer from chest pain and decreased exercise duration at COHb levels measured from 1% - 9% .
COHb levels between 30-70% lead to loss of consciousness and eventually death .
Long-term Effects
Following the resolution of the acute symptoms, there may be a lucid interval from 2-40 days before the development of delayed neurologic sequelae (DNS). Diffuse brain demyelination combined with lethargy, behavioral changes, memory loss, and parkinsonian features may occur. 75% of patients with DNS recover within 1 year. Neuropsychologic abnormalities with chronic CO exposure are found even when magnetic resonance imaging (MRI) and magnetic resonance spectroscopy findings are normal. White-matter damage in the centrum semiovale and periventricular area of the brain, and abnormalities in the globus pallidus, are most commonly observed on MRI following CO toxicity. Though less common, toxic or ischemic peripheral neuropathies are associated with high levels of CO exposure in humans. The basis for the management of CO poisoning is 100% hyperbaric oxygen therapy using a tight-fitting mask for at least 6 hours. The indications for treatment with hyperbaric oxygen to decrease the half-life of COHb remain controversial .
Description
Carbon monoxide is a colorless, odorless, tasteless, flammable, toxic gas.Carbon monoxide is produced when carbon and carbon compounds undergo incomplete combustion. The inefficient combustion of carbon fuels for heating results in the production of carbon monoxide, which may result in high CO concentrations in indoor environments. The use of carbon fuel heaters without adequate ventilation can result in deadly conditions. Each year several hundred people in the United States die from CO poisoning, and 10,000 patients are treated in hospitals for CO exposure.Cars and other forms of transportation are a major source of carbon monoxide pollution in cities.
Description
Carbon monoxide, produced by the incomplete combustion of organic materials, comes from automobile exhaust, tobacco smoke, and malfunctioning gas heaters. It has a greater affinity for hemoglobin in the bloodstream than does oxygen, and several hundred parts per million in the air can cause symptoms of oxygen deprivation.
The Uses of CARBON MONOXIDE
Carbon monoxide is used in the oxo processor Fischer–Tropsch process in the produc tion of synthetic fuel gas (producer gas, watergas, etc.); as a reducing agent in the Monodprocess for the recovery of nickel; in car bonylation reactions; and in the productionof metal carbonyls and complexes. It is pro duced by incomplete combustion of organicmaterials. Risk of exposure to this gas arisesunder fire conditions; from a burning stoveor from burning wood or candles in a closedroom; in the exhausts of internal combustion engines; in a closed garage with theautoengine on; and from oil or gas burners,improperly adjusted.
Indications
Used as a marker of respiratory status in spirometry tests , .
Food additive for pigment fixation in meat .
Background
Carbon monoxide (CO) is a colorless, odorless, and tasteless gas that has a slightly lower density than air. It is toxic to hemoglobin utilizing animals (including humans), when encountered in concentrations above about 35 ppm, although it is also formed in normal animal metabolism in low quantities, and is thought to have some normal biological/homeostatic functions . Carbon monoxide (CO), is a ubiquitous environmental product of organic combustion, which is also formed endogenously in the human body, as the byproduct of heme metabolism . Exhaled CO (eCO), similar to exhaled nitric oxide (eNO), has been evaluated as a candidate breath biomarker of pathophysiological states, including smoking status, and inflammatory diseases of the lung and other organs. Exhalation of corbon monoxide values have been studied as potential indicators of inflammation in asthma, stable COPD and exacerbations, cystic fibrosis, lung cancer, and during surgery or critical care .
A test of the diffusing capacity of the lungs for carbon monoxide (DLCO), is one of the most clinically valuable tests of lung function testing. The technique was first described 100 years ago, and applied to clinical practice many years after. The DLCO measures the ability of the lungs to transfer gas from inhaled air to the red blood cells in pulmonary capillaries. The DLCO test is both convenient and simple for the patient to undergo. The ten seconds of breath-holding required for the DLCO maneuver is easier for most patients to perform than the forced exhalation required for other respiratory tests .
Carbon monoxide is presently used in small amounts in low oxygen modified atmosphere packaging systems (MAP) for fresh meat to stabilize and maintain natural meat color. This use of CO has been generally recognized as safe (GRAS) in several packaging applications for fresh meat products. Since 2002, FDA has favorably reviewed three GRAS notifications for carbon monoxide use in fresh meat packaging . The FDA classifies this drug as permitted as a food additive in the packaging and preparation of food products, while following the federal code of regulations .
There have been several concerns voiced of over the use of carbon monoxide in food products , , . The European Union has banned the use of carbon monoxide as a color stabilizer in meat and fish. A December 2001 report from the European Commission's Scientific Committee on Food concluded that the gas did not pose a risk provided that food was maintained adequately cold during storage and transport to prevent the growth of microorganisms . In New Zealand, the use of carbon monoxide in fish preparation has been banned, as it may mask the effects of food spoilage and bacterial growth .
Pharmacokinetics
Carbon monoxide is used to measure the diffusing capacity for carbon monoxide (DLCO), also known as the transfer factor for carbon monoxide. It is a measure of the gas transfer from inspired gas to the circulatory system (red blood cells in particular) . It is used in a particular pulmonary function test called "the single-breath test" .
DLCO, measured for clinical and research purposes almost exclusively by the single-breath method is an important and very useful pulmonary function test. It is useful in the evaluation of patients with dyspnea, obstructive lung diseases, restrictive lung diseases, and in patients with diseases of the pulmonary vasculature. The measurement of DLCO using carbon monoxide is representative of the surface area available, the volume of blood present in the pulmonary capillaries, as well as the thickness of the alveolar-capillary membrane .
Conditions that increase DLCO: Heart failure, erythrocythemia, alveolar hemorrhage, asthma
Conditions that decrease DLCO: emphysema, pulmonary fibrosis, pulmonary hypertension, pulmonary embolism
In addition to the above uses, carbon monoxide (CO) is increasingly being accepted in recent years as a protective molecule with important signaling capabilities in both physiological/homeostatic and pathophysiological situations. The endogenous production of CO occurs via the activity of constitutive (heme oxygenase 2) and inducible (heme oxygenase 1) heme oxygenase enzymes, which are both responsible for the breakdown of heme. Through the generation of its products, which in addition to carbon monoxide, includes the biliary pigments biliverdin, bilirubin and ferrous iron, the heme oxygenase 1 system also have an essential role in the regulation of the stress response and in cell adaptation to injury. Preclinical studies have suggested potential benefits of carbon monoxide in cardiovascular disease, inflammatory disorders, as well as organ transplantation .
Metabolism
Not Available
Properties of CARBON MONOXIDE
| Melting point: | −205 °C(lit.) |
| Boiling point: | −191.5 °C(lit.) |
| Density | d4-195 (liq) 0.814; d (gas) 0.968 (air = 1.000); d40 at 760 mm: 1.250 g/liter |
| solubility | At 20 °C and at a pressure of 101 kPa, 2.266 volumes of carbon monoxide dissolve in 100 volumes of water. |
| appearance | Colorless gas |
| form | colorless gas |
| Odor | Odorless gas |
| Water Solubility | mL/100mL H2O: 3.3 (0°C), 2.3 (20°C) [MER06] |
Safety information for CARBON MONOXIDE
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Gas Cylinder Compressed Gases GHS04  Skull and Crossbones Acute Toxicity GHS06  Health Hazard GHS08 |
| GHS Hazard Statements |
H220:Flammable gases H280:Gases under pressure H331:Acute toxicity,inhalation H372:Specific target organ toxicity, repeated exposure |
| Precautionary Statement Codes |
P202:Do not handle until all safety precautions have been read and understood. P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P308+P313:IF exposed or concerned: Get medical advice/attention. P403+P233:Store in a well-ventilated place. Keep container tightly closed. P410+P403:Protect from sunlight. Store in a well-ventilated place. |
Computed Descriptors for CARBON MONOXIDE
| InChIKey | UGFAIRIUMAVXCW-UHFFFAOYSA-N |
Abamectin manufacturer
Maharashtra Gas Co
INOX Air Products Pvt Ltd
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol 3-NITRO-5-ACETYL IMINODIBENZYL (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran

You may like
-
 630-08-0 Carbon monoxide 98%View Details
630-08-0 Carbon monoxide 98%View Details
630-08-0 -
 630-08-0 98%View Details
630-08-0 98%View Details
630-08-0 -
 Carbon monoxide 99%View Details
Carbon monoxide 99%View Details
630-08-0 -
 68915-31-1 99%View Details
68915-31-1 99%View Details
68915-31-1 -
 Azadirachtin 11141-17-6 99%View Details
Azadirachtin 11141-17-6 99%View Details
11141-17-6 -
 Geraniol 99%View Details
Geraniol 99%View Details
106-24-1 -
 BENZALKONIUM CHLORIDE BKC 99%View Details
BENZALKONIUM CHLORIDE BKC 99%View Details
8001-54-5 -
 Amrit Neem 11141-17-6 99%View Details
Amrit Neem 11141-17-6 99%View Details
11141-17-6